Introduction
The Valsalva maneuver is a widely used physiologic technique for the non-invasive evaluation of heart murmurs and ventricular function [1-3]. The Valsalva maneuver consists of forceful expiration against a closed glottis, resulting in an increase in both intra-thoracic and intra-abdominal pressure, and activation of autonomic nervous function [4-6]. Although the hemodynamic changes during Valsalva maneuver have been well documented, these have been focused on cardiac chambers, aorta and systemic large veins [4,7-10]. Anatomically, venous return consists of systemic and hepatic venous return, and systemic venous return decreased markedly during the Valsalva maneuver [8,11]. However, it was not well defined that hemodynamic changes focused on the liver during Valsalva maneuver. Clinically, the Valsalva maneuver is considered as main cause of defecation syncope and surgery performed in a patient with functional suprahepatic inferior vana cava (IVC) obstruction during the Valsalva maneuver [12]. Collapsed IVC showed during the maneuver in normal healthy subjects using ultrasonography study, but IVC showed angular appearance and not collapsed during the maneuver in the venography study [10,11]. The change of IVC during the maneuver is ambiguous and hemodynamic contribution of hepatic vein and portal vein during the maneuver has not been studied in normal healthy subjects.
Duplex Doppler ultrasonography of the liver provides important information about liver condition [11,13]. Hepatic vein flow depends on hepatic parenchymal compliance, thoracoabdominal pressure, and right atrial pressure. It is known that hepatic vein Doppler waveform is triphasic pattern which is composed of two anterograde flow peaks toward the heart and one retrograde flow peak toward the liver in healthy subjects [14]. Recently, volume flow measurement has been used for quantification of blood flow and it showed good correlation with magnetic resonance in quantification of cerebral blood flow [15-17]. Therefore, analysis of flow pattern and quantification of liver flow can be helpful to understand liver condition under the Valsalva maneuver. Even the liver hemodynamic changes during the Valsalva maneuver in healthy volunteers have been studied in previous reports, its contribution to venous return is not focused enough [18,19]. We hypothesized that the hepatic circulation might be an important role to maintain venous return to the heart during Valsalva maneuver. The aim of our study was to assess the hemodynamic change of liver including hepatic vein and portal vein during the Valsalva maneuver.
Materials and Methods
Subjects
Institutional Review Board was obtained by our institute. For this prospective study, volunteers who had no known liver or heart disease were included. Volunteers with heart diseases were excluded by medical history, and those with diffuse liver parenchymal diseases were also excluded by clinical, laboratory, and B-mode ultrasonography. To prevent the effects of obesity in the evaluation, volunteers with body mass index over 25 were excluded. Volunteers with a previous history of abdominal or thoracic surgery or alcohol use or taking cardiovascular medication that may have an adverse effect on the liver were also disqualified to precluded possible influences on the waveform of the hepatic vein. Totally 30 consecutive adults with sufficient inclusion criteria participated in our study. They were men, with a mean age of 25.5±3.64 years (range, 22 to 33 years), who were gathered from military service. The volunteers were briefed about the study protocol, and then written consents for this study were obtained.
Ultrasonography and Analysis
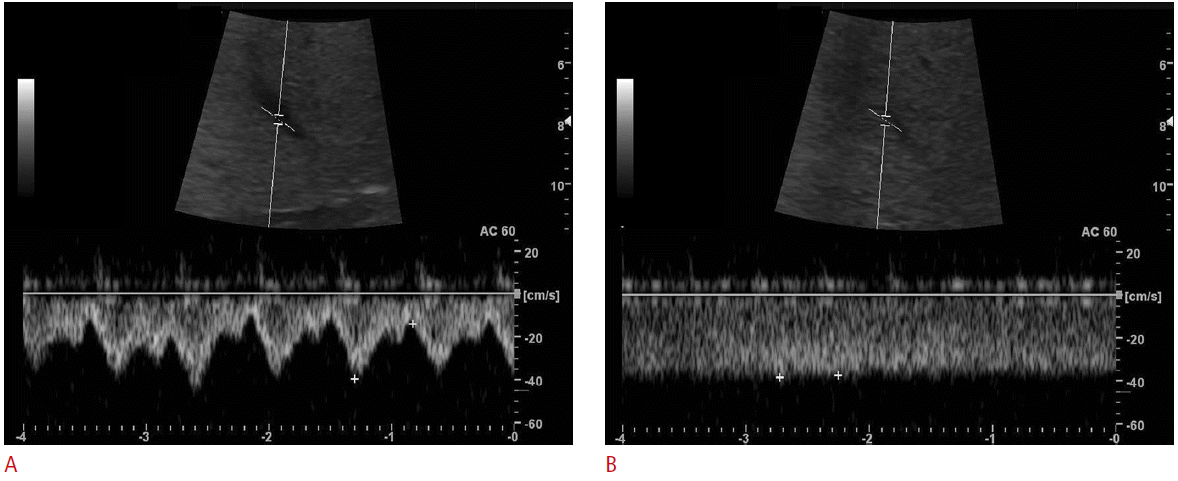
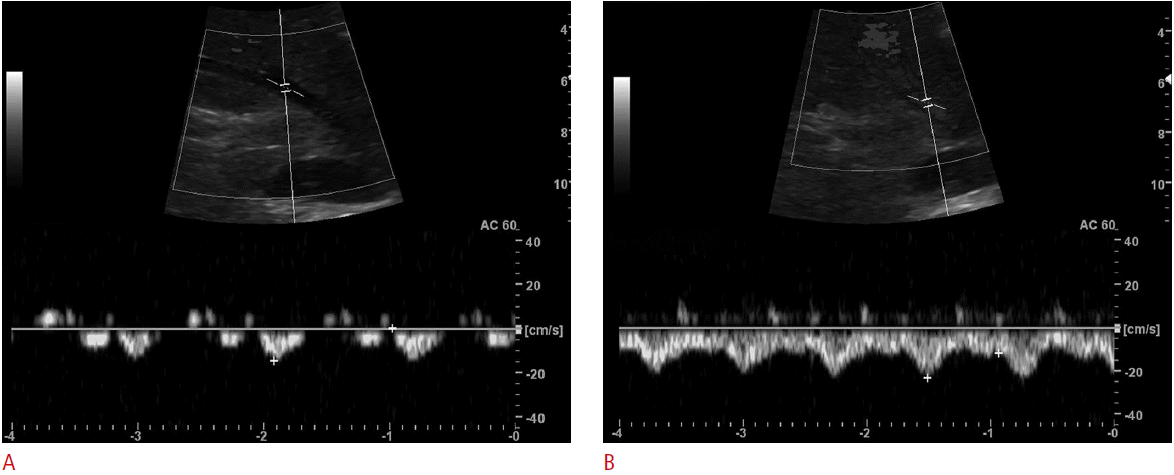
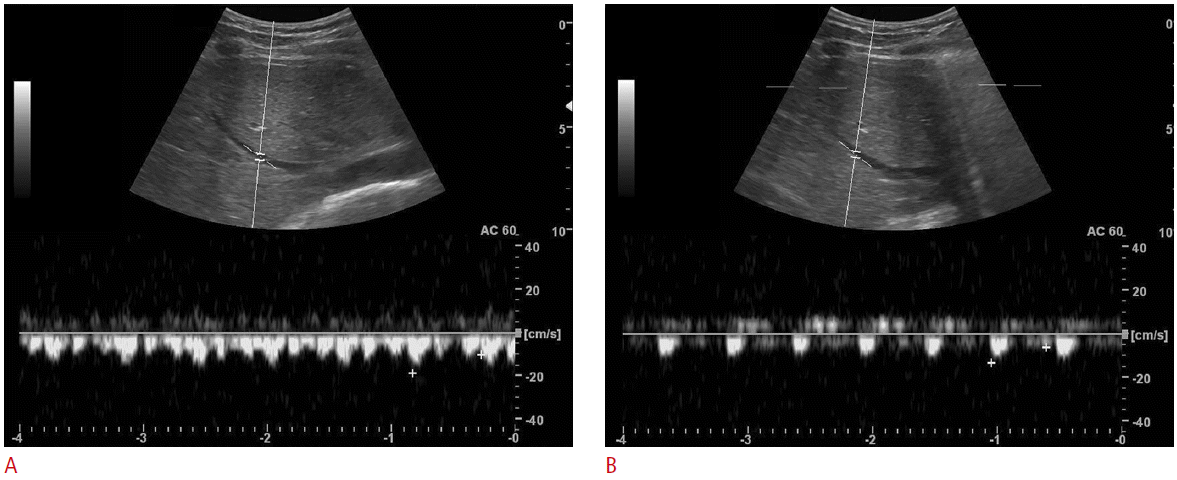
The subjects underwent ultrasonography in the sitting position. The ultrasonography was performed by a radiologist (DHB) using 4.0 MHz convex probe (LOGIQ 9, GE Healthcare, Waukesha, WI, USA). Before evaluation of the vascular flow was performed, a routine examination of the liver was made to exclude any pathologic processes. Subjects underwent ultrasonography during both rest and Valsalva maneuver. The subjects were instructed to perform Valsalva maneuver, which were full inspiration followed closed glottis with forceful expiration for 10-20 seconds. Scanning parameters were as follows: depth of the display, 16.0 cm; one focal zone at the target vessel, dynamic range, 75 dB; frame rate, 31 Hz; time gain control at its central position, and power at a constant level. For ultrasonography at rest, all subjects were asked to breathe shallowly while the images were captured in order to facilitate good quality images. The mid-portion of right main portal vein (RMPV) was imaged on behalf of portal veins and the nearest confluent portion of middle hepatic vein (MHV) was imaged on behalf of hepatic veins, because these vessels were adequate for Doppler ultrasonography. Ultrasonograms of the intrahepatic IVC and MHV were obtained by a subcostal approach and intercostal approach was used for RMPV imaging. Spectral Doppler ultrasonography was used for measure of vascular flow of the RMPV and MHV. The waveforms of a MHV were classified into three groups: triphasic waveform, biphasic waveform without a reverse flow, and monophasic waveform. Maximal and minimal velocities of the RMPV and MHV were measured. For flowvolume measurements, the RMPV and MHV were targeted. The same longitudinal plane along the target vessels was used for the flow measurements, maintaining the plane of section as constant as possible. Windows of spectral Doppler were used to place the sample volume for supplemental pulsed wave interrogation at the selected site of measurement. The sample volume was within the vessel diameter and changed during the Valsalva maneuver because of diameter of MHV decreased. Angle of insonation was adjusted at or below 60° and was kept constant while both rest and Valsalva maneuver.
The luminal diameter for measurement of volume flow was measured on the magnified images of the vessel. The volume flow measurements were automatically calculated by the built-in software of the ultrasonography device after the image was frozen, as the product of angle-corrected, time-averaged flow velocity, and the diameter of the vessels. The luminal diameter and flow measurements were repeated 3 times for each vessel and then averaged to provide the value estimates of each vessel to minimize the random errors to an acceptable level. All ultrasonography was performed by a single radiologist, experienced in ultrasonography.
Statistical Analysis
Quantitative analysis was performed by the same radiologist who performed ultrasonography. The diameter of intrahepatic IVC, and diameter, and minimal and maximal velocities and volume flow of the MHV and RMPV at rest were compared to those during the Valsalva maneuver using paired t-test. Statistical significance was set at P<0.05. The analysis was performed using SPSS ver. 12.0 (SPSS Inc., Chicago, IL, USA).
Results
During the Valsalva maneuver, the diameters of the intrahepatic IVC decreased. The half of all subjects’ intrahepatic IVC collapsed (n=15) and other half showed decrease in diameter (n=15). The mean diameters (cm) of the intrahepatic IVC at rest and Valsalva maneuver were 1.94±0.40 versus 0.56±0.66 (P<0.001). The mean diameter (cm), minimal velocity (cm/sec), maximal velocity (cm/sec), and volume flow (mL/min) of MHV at rest and Valsalva maneuver were 0.60±0.15 versus 0.38±0.20 (P<0.001), -7.98±5.47 versus 25.74±13.13 (P<0.001), 21.34±6.89 versus 35.12±19.95 (P=0.002), and 106.94±97.65 versus 153.90±151.80 (P=0.014), respectively. The mean diameter (cm), minimal velocity (cm/sec), maximal velocity (cm/sec), and volume flow (mL/min) of RMPV at rest and Valsalva maneuver were 0.78±0.21 versus 0.76±0.20 (P=0.485), 20.21±8.22 versus 18.73±7.43 (P=0.351), 26.79±8.85 versus 24.93±9.91 (P=0.275), and 391.52±265.63 versus 378.43±239.36 (P=0.315), respectively (Table 1).
Discussion
Anatomically, the hemodynamics of liver can be classified as influx to the liver and efflux from the liver. Influx system includes hepatic artery and portal vein, efflux system is hepatic vein. Even though the hepatic artery was not assessed by spectral Doppler ultrasonography due to its narrow diameter, change of hepatic artery flow might be minimal or increased during Valsalva maneuver [20]. Therefore, hemodynamic influx from the portal vein and the hepatic artery to the liver may maintain or slightly increase during the maneuver. In normal condition, hepatic influx flow are same to hepatic efflux flow. That suggests that increased hepatic venous flow lead to increased portal venous or hepatic arterial flow. However, our data showed that reciprocal result of increased hepatic venous flow and decreased portal flow even though it was not statistically significant during Valsalva maneuver. This results might be due to ultrasonography observation for short time interval of 10-20 seconds during the Valsalva maneuver. Rather than this point, we suggest that increased hepatic venous flow was temporal compensatory effect for decreased systemic venous return by direct external compression of the inferior vena cava during the Valsalva maneuver. In addition, there is a wide variation in venous return from subject to subject, produced by a wide variation in splanchnic pooling from subject to subject during the Valsalva maneuver [21].
Under those conditions, Valsalva maneuver increases intraabdominal pressure, so the liver may be squeezed, and the hepatic vein is simultaneously squeezed. In our study, the diameter of the hepatic vein decreased, minimal and maximal velocities and volume flow of the hepatic vein increased. Besides squeezing effect of Valsalva maneuver to hepatic vein, increased abdominal pressure increase intrahepatic pressure and make pressure gradient between the liver and heart. Blood flow is determined by pressure gradient. The pressure gradient maintained during Valsalva maneuver and can produce stable venous return to the heart during the maneuver. When Valsalva maneuver finishes, the increased intra-abdominal pressure and thoracic pressure are relaxed. At that time, venous return through the liver would suddenly decrease, and venous return through the systemic large veins suddenly increases. This finding indicated that increased hepatic venous flow during the Valsalva maneuver was not directly related to decreased systemic venous return, but it would be a function of maintenance of venous return to the heart by squeezing effect of liver.
Doppler waveform of hepatic vein is determined by the balance between hepatic pressure and right atrial pressure. Valsalva maneuver can elevate intra-abdominal pressure, its effect on the liver can be various. On the other hand, during ventricular systole, the tricuspid annulus moves toward the cardiac apex, causing suction of blood into the right atrium from the liver. The result is a dominant antegrade wave [18]. Mediastinal volume decreases during the Valsalva maneuver, which may limit cardiac motion, so the suction effect by ventricular contraction may be reduced. This effect may be various. The monophasic pattern seems to be seen in subjects with effectively increased hepatic pressure representing as high baseline hepatic vein velocity and limited cardiac motion, and it showed in 80% of all subjects in our study. A biphasic pattern seems to be seen in subjects with insufficiently increased hepatic pressure and insufficiently limited cardiac motion. One subject who did not show the waveform changes in the hepatic vein during the Valsalva maneuver in our study. We think that the subject may fail to increase hepatic pressure.
A collapsed intrahepatic IVC was also an important finding in our study. To evaluate hepatic hemodynamic changes during Valsalva maneuver is important because there is no confirmative study that functional IVC stenosis should be treated by surgery. Anatomically, IVC can be divided as suprahepatic portion, intrahepatic portion, and infrahepatic portion. In a previous report, the patient who had defecation syncope with functional stenosis at suprahepatic portion was corrected by surgery [12]. This seems to be reasonable because suprahepatic IVC stenosis may be vulnerable to take sufficient venous return during Valsalva maneuver. However, intrahepatic portion IVC stenosis does not seems to be surgical disease on the basis of our results. In previous studies, the IVC changes during Valsalva maneuver were ambiguous [10,11]. The IVC was collapsed during Valsalva maneuver in a study with ultrasonography and the IVC was intrahepatic portion [11]. However, the IVC was not collapsed during Valsalva maneuver in other study with venography [10]. We evaluate intrahepatic portion of IVC and it showed decreased in diameter in half of the subjects and collapsed in other half of the subjects. Venography study might be influenced by injector with pressure, so liver ultrasonography seems to be better except for lack of direct velocity measurement of IVC. On the basis of our study, hepatic vein might have an important role to maintain venous return during Valsalva maneuver. Therefore, in our knowledge, functional IVC stenosis may be treated by surgery if the stenotic site is at suprahepatic portion. However, if the stenotic site is at intrahepatic portion or infrahepatic portion, functional IVC stenosis should not be treated by surgery. Those cases may be physiologic findings during Valsalva maneuver. Further study might be necessary to assess the hemodynamic change of liver in patients with defecation syncope.
Our study had some limitations. First, the study group was too small to figure out the effect of Valsalva maneuver and the volunteers included in our study were young healthy males. We thought that obesity could increase squeezing effect of the Valsalva maneuver and was one of exclusion criteria. Therefore, our data might be skewed to very healthy young men. However, our study suggests that the hepatic vein Doppler waveform be different in each subject during the Valsalva maneuver. Second, we did not assess the patient groups of liver cirrhosis or defecation syncope in our study. Our study focused on hepatic hemodynamics during Valsalva maneuver in healthy subjects which have not been focused enough. Third, we could not obtain all portal veins and hepatic veins because of long acquisition time and inadequate angle for Doppler ultrasonography. In addition, we could not evaluate the changes of temporally increased of hepatic venous efflux, which would be same to hepatic influx due to decreased volume of liver by squeezing effect during the Valsalva maneuver. It was a physiologically inevitable limitation because the Valsalva maneuver could not be maintained for a long time. Fourth, we could not use manometry to quantify the Valsalva maneuver and could not measure intra-thoracic and intra-abdominal pressure because of lacking equipment. However, we educated the subjects in our study before the ultrasonography and checked the Valsalva maneuver as strong as they could do. Fifth, we assessed MHV flow, which may be different from other hepatic vein flow because of different hepatic mass around the vessel. Sixth, there were technical problems to perform spectral Doppler during Valsalva maneuver. Practically, it was difficult to evaluate MHV velocity and volume flow because its diameter decreased markedly and its location changed during the maneuver.
In conclusion, hepatic hemodynamics during Valsalva maneuver are presented in our study. Flow velocity and volume flow of hepatic vein were increased, but those of portal vein were not affected by Valsalva maneuver. These findings suggest the temporally increased hepatic venous return by squeezing effect of liver might play an important role to maintain systemic venous return during Valsalva maneuver.



 Print
Print facebook
facebook twitter
twitter Linkedin
Linkedin google+
google+
 Download Citation
Download Citation PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC




