Introduction
Nodular goiters and thyroid nodules are clinically palpable in about 5% of the general population. When visualized using ultrasonography, the prevalence of thyroid nodules is even higher, with reported rates as high as 60% [1,2]. Fortunately, despite the high prevalence, most of these nodules are benign in nature and will remain static over time; as such, no active intervention is required [3]. However, up to 10%-15% of these nodules can progress and grow, and some can even cause obstructive symptoms [4]. In such circumstances, surgery is indicated [2]. However, surgery results in morbidity and requires general anesthesia and hospitalization [5]. As a result, there has been an increasing interest in developing less invasive, non-surgical techniques for treating sizable or growing thyroid nodules [6]. For predominantly-solid or solid nodules, image-guided thermal ablation techniques such as laser ablation (LA), microwave ablation, and radiofrequency ablation (RFA) are the most established [7,8]. They have been shown to be effective not only in causing physical shrinkage, but also in improving nodule-related symptoms.
High-intensity focused ultrasound (HIFU) is a relatively new ablation technique that has been less thoroughly described. It utilizes focused ultrasonic energy to induce tissue ablation, and therefore does not require the insertion of a needle or probe through the skin. Like other ablation techniques, it is effective in causing significant nodule shrinkage and rapidly alleviating obstructive symptoms [9-11]. Although the longer-term outcomes of this modality remain unclear, there is an increasing interest in this technique. Therefore, it would be timely to examine the current development and future directions of this technology for the treatment of thyroid diseases.
Focused Ultrasound Technology
The concept of using ultrasonic energy as a penetrating force to destroy tissue at a distance originated in the last century and was summarized by Kremkau in the 1970s [12]. In the 1950s, researchers such as Barnard et al. [13] and Fry and colleagues [14-16] from Illinois in the United States applied ultrasonic energy in a clinical setting by treating Parkinson disease and other neurological conditions with focused ultrasound energy. However, it was not until the 1990s that interest in applying HIFU therapy in the treatment of solid tumors emerged again.
HIFU is based on the unique characteristic of sound waves between the frequencies of 0.8 to 3.5 MHz, which can be intensified to focus at a distance from the radiating transducer. The harnessed acoustic energy (up to 10,000 W/cm2) causes intense vibrations of tissue, generating heat that causes necrosis at the target location without collateral damage to the surrounding structures. The benefit of inducing immediate tissue death from a distance by an ultrasonic energy source without the need of invasive surgery or insertion of ablation instruments makes HIFU an attractive therapeutic option. Although data on the efficacy of HIFU are still being gathered, the initial results obtained from pioneering researchers in Asia have been encouraging regarding the treatment of solid tumors, such as those in the liver and kidneys [17-23].
The mechanism of injury induced by HIFU is two-fold, thermal and mechanical. In thermal injury, injury occurs as a result of maintaining a high temperature within the target. A temperature of 56℃ or above causes protein denaturation and coagulative necrosis in seconds [24]. At this temperature, water within body tissues starts to vaporize, and microbubbles begin to form within the target. Mechanical injury, in contrast, is related to the process of cavitation [25,26]. This is a process where bubbles develop and enlarge within the target. As multiple bubbles expand and collapse, mechanical damage to nearby cells occurs. The popcorn effect is the typical example of cavitation [25]. Through these mechanisms, irreversible cell death occurs via coagulative necrosis and apoptosis.
Other important biological effects of HIFU also take place after treatment. Cells undergo subtle changes in response to exposure to ultrasonic energy, resulting in cell arrest. Cellular membranes also become more permeable, leading to changes in the intracellular milieu [26]. This effect on intracellular proteins leads to cell arrest. The resultant cell death triggers the activation of immune cells such as natural killer cells, CD8+ T cells, and dendritic cells, which roam the bloodstream to detect and destroy tumor cells both locally and systemically [26].
Since the HIFU energy is concentrated in a small area, the damage to the overlying structures is minimal and the temperature outside the focal point remains low. This property helps to minimize inadvertent collateral damage.
Current Devices and Procedures
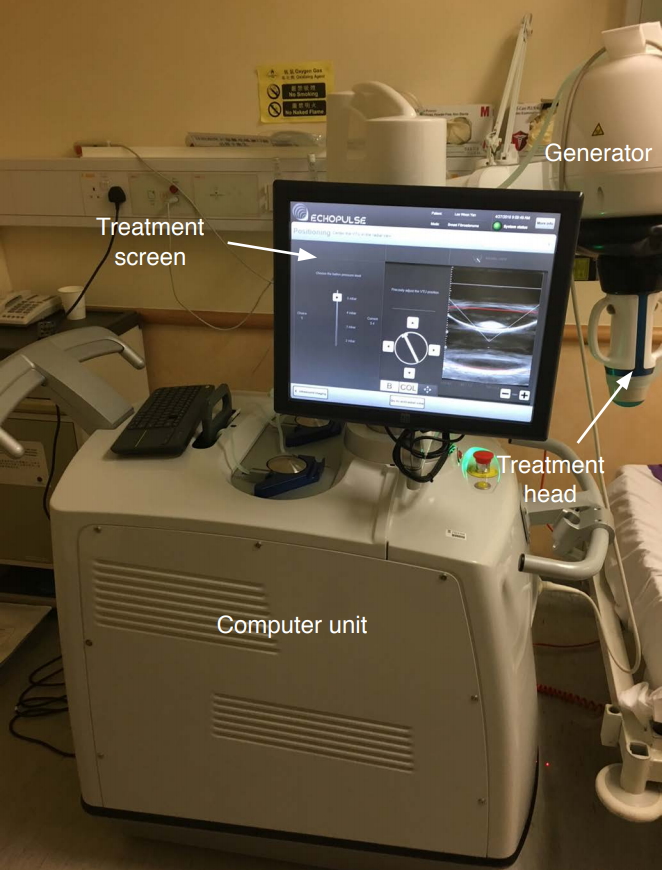
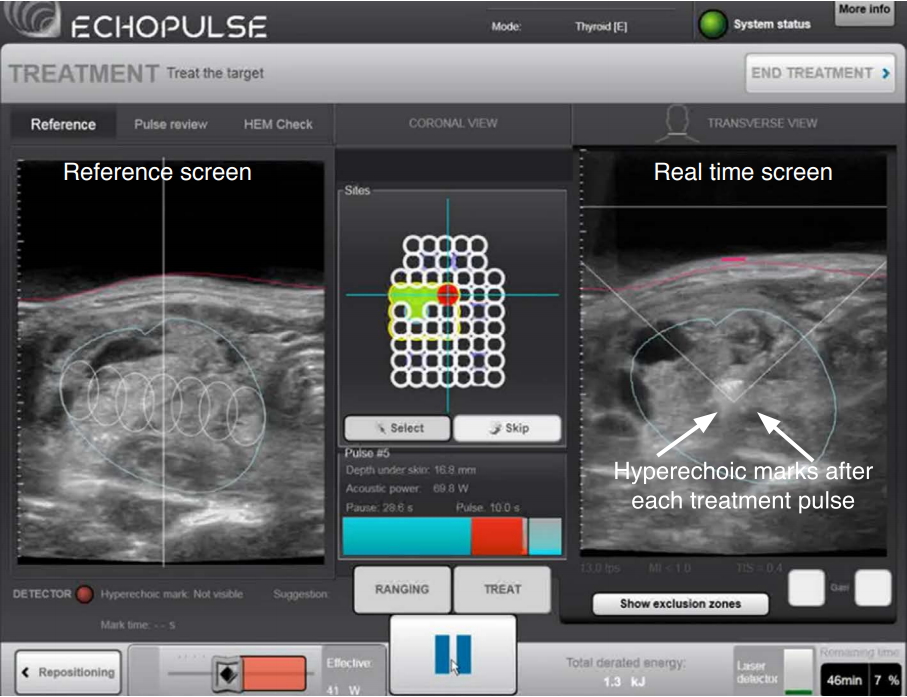
To date, only one commercial device has been approved and is available for the ablation of thyroid nodules. The procedure is done entirely under ultrasound guidance. The device itself has an energy generator, a treatment head, a skin cooling device, and a touch-screen interface for planning (Fig. 1). The treatment head incorporates an image transducer (7.5 MHz, 128 elements, linear array) and an HIFU transducer (3 MHz, single element, 60 mm in diameter). Before the treatment starts, the treatment head is placed on the neck on the side of the index nodule and is finely adjusted until the nodule is within the treatable depth of 7 to 30 mm from the skin surface. Once marked on the treatment screen, the device’s computer automatically divides the nodule into multiple ablation voxels. Each voxel measures approximately 7.3 mm in thickness and 5 mm in width and receives a continuous 8-second pulse of HIFU energy followed by 20-30 seconds of cooling time before the beam moves on to the next voxel (Fig. 2). To ensure safety, nearby structures such as the carotid artery, trachea, and skin are marked on the treatment screen before the start of treatment by the operator [27-29]. To avoid inadvertent heat injury to important surrounding structures, the device automatically selects safety margins for the skin, the trachea, and the ipsilateral carotid artery. A laser-based movement detector enables immediate power interruption when the patient moves or swallows during ablation. To avoid skin burn, the skin is cooled by a balloon (filled with 10℃ liquid) at the tip of the treatment head. Both the total amount of energy delivered to the nodule (in kilojoules) and the "on-beam" (sonification) time taken (in minutes) are automatically recorded by the device's computer. The on-beam treatment time is the duration from the first to the last pulse (in minutes). On average, the total onbeam time for a 3-cm nodule ranges from 30 to 45 minutes [29-32], while for larger-sized nodules (i.e., with a volume >30 mL), two or more sequential treatments might be necessary in order to achieve complete ablation. In comparison to other, more established ablation techniques such as RFA or LA, HIFU generally takes almost twice as long. However, this problem can be resolved by shortening the cooling period between each treatment pulse with new ablation software. An oral diet is resumed immediately afterwards and patients can be discharged home 2-3 hours after treatment.
Pain Control during Treatment
Good pain control is an important aspect of ensuring successful HIFU treatment, because pain can often lead to incomplete or suboptimal ablation of the target. Although the exact source or origin of the pain is unclear, pain is likely to be felt as high levels of ultrasound energy travel from the skin into the target. To alleviate some of this pain, a local infiltration of 1% lidocaine around the thyroid gland is often used. In addition, we often administer intravenous pethidine and diazepam boluses at the start of treatment. To date, this combination has worked well, and up to 15% of patients reported experiencing absolutely no pain during treatment [30]. For more posteriorly located nodules, some patients may complain of pain or discomfort arising from the back of the neck and shoulder areas. This might occur because some of the HIFU energy may travel through the thyroid gland into the paraspinal muscles beneath the pre-vertebral fascia. To alleviate this, the operator can reduce the treatment depth or reduce the power of each treatment pulse. Interestingly, a research group has recently described the feasibility of low-energy HIFU ablation in thyroid nodules without the need for any anesthesia or analgesia [11]. Therefore, it is possible to deliver good ablation without anesthesia or analgesia by setting the power of each treatment pulse to its minimum setting.
Short-Term Efficacy
In terms of treatment efficacy following single treatments, HIFU can generally achieve a volume shrinkage of the nodule between 45% to 70%, depending on the initial size of the nodule (Fig. 3) [9-11,28]. In our experience, small-sized nodules tend to respond better than large-sized nodules to single rounds of treatment. A previous study showed that for nodules larger than 30 mL, the mean 6-month volume reduction was 48%, while for intermediate-sized nodules (10-30 mL), the mean volume reduction was 68% [27]. For small nodules (less than 10 mL), the mean volume reduction was 78% [27]. However, it is worth pointing out that there is no well-defined upper limit for HIFU ablation, and single ablations can certainly be applied to nodules measuring up to 5 cm in diameter [27]. A strategy proposed to achieve better efficacy is to perform two sequential treatments of the same nodule. However, this strategy has yet to be evaluated in the literature.
Apart from the physical shrinkage of nodules, it is worth highlighting that HIFU leads to improvements in the severity of pressure symptoms arising from nodules over a period of 12 months. Relative to patients who were managed expectantly, at 12 months, patients who underwent HIFU ablation reported significantly fewer pressure symptoms and better physical composite scores (as measured by a visual analogue scale [VAS] with scores of 0-10 and a standard health-related quality of life tool, respectively) [10]. Interestingly, there was a significant correlation between physical composite scores and 6- and 12-month reductions in nodule size [10].
Treatment-Related Risks
Vocal cord paresis (VCP) is defined as a reduction or absence of movement in one or both of the vocal cords. It is the most common complication following HIFU ablation of thyroid nodules. The incidence of VCP after HIFU has been reported to be approximately 1%-2% when the vocal cords are examined routinely after each procedure. The reason for its occurrence is because the recurrent laryngeal nerve runs closely behind the thyroid lobe in the tracheoesophageal groove, and if the nodule (or target) is located near the posterior thyroid capsule, close to the tracheoesophageal groove, either the heat or the HIFU wave itself can easily damage the nerve, resulting in VCP. This proposal is supported by a previous study that found the distance between the focus of the HIFU beam and the tracheoesophageal groove to be significantly related to the risk of VCP. To avoid VCP, a safe distance of 1.1 cm between the focus of the beam and the tracheoesophageal groove is essential [29].
Other complications, such as inadvertent skin burns and hypothyroidism, are rare. To avoid burning the skin, special attention must be paid during treatment to possible changes of the tumor location caused by the patient’s movement. Overshooting or undershooting of the nodule by the HIFU beam can cause inadvertent heat damage to the skin or deeper neck structures. A rise in the temperature of the skin surface may occur, and regular monitoring of skin changes is therefore crucial for preventing thermal injury. Fortunately, skin burns after HIFU ablation are extremely rare (<1%) when precautions have been taken. An additional safety mechanism is installed to interrupt the energy delivery in case of sudden body movements in order to decrease the incidence and severity of skin burns. Hypothyroidism as a direct result of HIFU ablation is very uncommon because the beam is normally focused at the center of the target, without damaging the surrounding functional parenchyma; therefore, the underlying thyroid function remains unaltered after treatment [31].
Long-Term Treatment Outcomes
Although HIFU is a promising form of ablation in the short-term, the medium- to long-term outcomes following a single HIFU treatment are not well established. In contrast to other ablation techniques such as RFA or LA, it is unclear whether the initial shrinkage from the HIFU ablation can persist beyond 12 months. Given that nodule regrowth can occur after thermal ablation, it is important to evaluate the longer-term outcomes of HIFU ablation. However, the current problem, of course, that the follow-up period for the treated nodules remains short and a longer follow-up period is required. To date, only a single study has reported the medium-term efficacy after single HIFU ablations of benign thyroid nodules. In that study, a total of 108 patients were analyzed and followed for 2 years. At 2 years, fewer than two-thirds (63/108, 58.3%) of nodules had a smaller volume (by >4.5%) than at 12 months, while about one-fifth of nodules (20.4%) had a small increase in volume compared to the volumes at 12 months [32]. However, the overall nodule shrinkage rates at 3, 6, 12, 18, and 24 months were 51.32%±20.71%, 62.99%±22.05%, 68.66%±18.48%, 69.76%±17.88%, and 70.41%±17.39%, respectively [32].
Role of a Second Round of HIFU Treatment
With greater experience, it has become clear that all thyroid nodules can shrink satisfactorily with a single treatment. In previous studies, approximately 20% of benign thyroid nodules failed to shrink by >50% in volume at 6 months [10,27]. Although surgical resection remains a viable treatment option when an ablated nodule has failed to shrink satisfactorily and is causing persistent obstructive symptoms, many patients still prefer a less invasive approach. The results of two or more treatments (reapplications) have been reported for other thermal ablation techniques [33,34], but this is less well-described for HIFU ablation. In a recent study of 28 patients who did not achieve 50% volume reduction at 6 months after the first HIFU treatment, the second treatment (or reapplication) of the same nodule yielded a further shrinkage (or volume reduction rate) of 21.78%±16.87% after 6 months. In short, the results of reapplication of HIFU ablation were not overwhelmingly encouraging. Nevertheless, the baseline symptom score (by VAS) (3.96±1.04) improved at 3 and 6 months after reapplication (2.96±1.43, P<0.001 and 2.58±1.39, P<0.001, respectively). Therefore, this option might still be worthwhile when a nodule has not shrunken after the first treatment. Interestingly, it was found that larger residual nodules experienced greater shrinkage than smaller residual nodules after the second treatment [35]. One reason for this might be that it is technically more difficult to focus on the un-ablated part of the nodule when it is small in volume.
Future Developments
There are still many unanswered questions related to the use of this technology in clinical practice. An active research area relates to the use of HIFU in the management of patients with relapsed Graves disease. The principle of this treatment is not dissimilar to that of subtotal thyroid resection, where the bulk of the functioning thyroid parenchyma is removed or de-functioned, leaving a smaller amount of viable thyroid tissue. The aim of the procedure involves thermal ablation of the center of both the right and left thyroid lobes, while leaving the peripheral parenchyma functional. In a pilot study, at 1 year, approximately 67% of patients remained free of anti-thyroid medications [36], and 3% experienced temporary vocal cord palsy. Therefore, HIFU may be a safe and efficacious treatment for patients with persistent or relapsed Graves disease. However, a randomized comparative study (between HIFU and other definitive treatments) is needed in order to recommend this treatment as a sound alternative for this condition.
Apart from Graves disease, other indications include locoregional recurrence of papillary thyroid carcinoma, as well as primary thyroid carcinoma. In a series of 39 patients, Lim et al. [37] tested HIFU for this indication by treating 61 recurrent papillary thyroid tumors and found that RFA could effectively control loco-regional recurrent papillary thyroid carcinoma without life-threatening complications; in other words, RFA may have a role in replacing "berry-picking surgery" in selected patients. Although considered to be the standard treatment for recurrence, repeated neck operations for recurrence are challenging and potentially hazardous due to the distortion of normal anatomy, which is replaced by adhesions and scar tissue, resulting in more morbidity, such as hypoparathyroidism and vocal cord palsy. Using ablation to treat recurrent cancer can be advantageous, since it is less invasive and can reduce complications. Another indication is for primary thyroid cancer. Since RFA is a potential treatment alternative for low-risk papillary thyroid microcarcinoma [38], it is certainly possible for HIFU to be used to treat small, low-risk papillary thyroid carcinomas.
Another area of research is the identification of early biomarkers or serum markers in predicting nodule shrinkage after ablation. At the moment, there is no way of knowing whether a nodule will shrink satisfactorily shortly after ablation. If a biomarker is available shortly after treatment, it could be possible to plan additional treatment earlier. In a previous study, the authors found that there was a significant rise in serum thyroglobulin (Tg) in the first few days after ablation. On average, the serum Tg level rose by 7-10 times in the first 4 days after ablation, suggesting that the degree of Tg elevation might be correlated with the extent of shrinkage [39]. More recently, it was found that a drop in anti-Tg levels on day 4 could also be used as a predictor of nodule shrinkage in patients with positive anti-Tg status [40].
With advances in technology, it is not unreasonable to expect better treatment results in terms of shortening the treatment time while maximizing the energy delivery. Although technological improvements fall outside the purview of physicians, increased experience with the modality will undoubtedly shorten the learning curve. One such recent advancement is the development of nanodroplets that, when injected intravenously, potentiate the effects of the ultrasonic energy. So far, experimental studies using intravenous nanodroplets have only been done in rat liver. However, the results are promising. HIFU-mediated liver damage was significantly enhanced with mixed perfluorocarbon nanodroplets when compared with HIFU alone [41]. When compared with a control arm of HIFU without an agent, the injection of nanodroplets achieved a 130% higher temperature and an ablated area that was 30 times greater than that of the control group [41]. This agent increased the ablation capacity by reducing the acoustic energy required to heat the lesion. While there was an increase in energy delivery, there was no corresponding increase in collateral damage to healthy tissue. In other words, the use of nanodroplets could reduce the procedure time without increasing the risk of burning the skin.
Finally, and perhaps most remarkably, there is an immunological aspect of HIFU that has gained increasing interest. Interest has emerged in using HIFU as an immune-modulating agent that might synergistically potentiate the effects of immunotherapy (like a programmed cell death ligand 1 inhibitor) in the treatment of metastatic cancers. As alluded to earlier regarding the immune response of cells to HIFU [24], cell death as a result of ablation triggers a cascade leading to downstream stimulation of the systemic response. This unexpected but powerful sequence of events can be manipulated to aid immunotherapy [42]. HIFU, a technique initially developed for local control, in fact has systemic ramifications. Thermal ablation may induce both regional and systemic production of cytokines through the activation of inflammatory cells. In studies using RFA, when compared with the control arm, serum interferon-gamma and vascular endothelial growth factor levels markedly increased after treatment [43]. In animal models, HIFU may modulate host antitumor immunity after the ablation of implanted neuroblastoma. The number of cytotoxic T lymphocytes (CTLs) and activated tumor-specific CTLs significantly increased. The same phenomenon has been observed in implanted colonic adenocarcinoma and melanoma [44]. In addition, dendritic cell infiltration of tumors was enhanced by HIFU ablation. A possible explanation is that the tumor left behind after ablation can induce a tumor-specific immune response, including the induction of CTL cytotoxic activity and enhanced activation of dendritic cells [44]. In essence, the residual cancer debris acts as an antigen for the immune system. The use of HIFU may reduce distant metastasis and potentially prevent local recurrence through activation of cancer patients’ immune system. Based on these findings, there is increasing interest in using HIFU and taking advantage of its immunological stimulation to enhance adjuvant immunotherapy. A phase I clinical trial is currently exploring the effects of HIFU on adjuvant immunotherapy. While the experiment is ongoing, the results of this prospective randomized controlled trial will have implications for the treatment protocol of metastatic solid tumors.
Conclusion
HIFU is a novel ablation technique that can cause effective shrinkage of thyroid nodules and alleviate pressure symptoms. Its main treatment indication remains benign thyroid nodules. Although the published results are promising, prospective studies with a longer follow-up period are required. Future possibilities include evaluating its role in Graves disease and low-risk malignant conditions, as well as exploring its synergistic effect with systemic immunotherapy.



 Print
Print facebook
facebook twitter
twitter Linkedin
Linkedin google+
google+

 Download Citation
Download Citation PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC




