Fetal tumors: prenatal ultrasonographic findings and clinical characteristics
Article information
Abstract
The incidence of fetal tumors has been increased due to generalization of prenatal evaluation and improvement of imaging techniques. The early detection of a fetal tumor and understanding of its imaging features are very important for fetal, maternal, and neonatal care. Ultrasonography is usually used for the detection and differential diagnosis of fetal tumors, and magnetic resonance imaging is increasingly being used as a complementary study. Many fetal tumors have different clinical and imaging features compared with pediatric tumors. Although several fetal tumors may mimic other common anomalies, some specific imaging features may carry early accurate diagnosis of fetal tumors, which may alter the prenatal management of a pregnancy and the mode of delivery, and facilitate immediate postnatal treatment.
Introduction
Although the prevalence of all congenital tumors was reported as the range of 1.7-13.5 per 100,000 live births, the true prevalence may be much higher because many cases of stillborn or aborted may not be reported [1-4]. Recent study reported higher incidence of congenital tumors [1], probably due to generalization of prenatal evaluation and improvement of imaging techniques.
The diagnosis of a fetal mass is a diagnostic and management dilemma because of the variety of possible differential diagnoses and the variable course during intrauterine and neonatal period [5,6]. The most challenging problem is to decide which case may be appropriate to ultrasonography (US) monitoring and postnatal surgery. The accurate prenatal diagnosis of a fetal tumor and the prediction for the need of prenatal intervention or immediate postnatal treatment is often impossible [5-8]. Many fetal tumors are not seen until relatively late during pregnancy and often as incidental findings in the late second or third trimester [7,9].
The prognosis of fetal tumor is generally poor, although there are some exceptions. Not only the histology of fetal tumors but also the location and size are important factors of the prognosis of fetal tumors. Some benign tumors are also fatal when tumors are large and causing cardiovascular compromise or airway obstruction.
The characteristics of fetal tumors are markedly different from those of pediatric tumors. Fetal tumors have different prevalence, locations, histologic features, and biologic behavior compared with pediatric tumors. The early detection of a fetal neoplasm and understanding of its imaging features are very important for fetal, maternal, and neonatal care. US is usually used as the first imaging modality for the detection and differential diagnosis of fetal tumors. Color Doppler US (CDUS) and Doppler spectrum analysis is helpful for understanding of the pathophysiology of fetal tumors and the accompanying fetal hemodynamic changes. Magnetic resonance imaging (MRI) is increasingly being used as a complementary study. The purpose of this review is to describe the prenatal imaging findings and clinical characteristics of relatively common fetal tumors categorized by their locations and origins.
Fetal Brain Tumors
Fetal intracranial tumors are rare, accounting for 0.5% to 1.9% of all pediatric tumors [10-14]. The most common fetal brain tumor is teratoma, followed by astrocystoma, craniopharyngioma, and primitive neuroectodermal tumor [15]. Fetal intracranial tumors are detected usually during the third trimester. Most are associated with a poor prognosis [10-15]. Such tumors can cause spontaneous intracranial hemorrhage or dystocia during delivery.
Teratoma is the most common congenital neoplasm composed of tissues originating from all three germinal layers, and may occur in a variety of locations. Fetal brain teratoma usually appears as a large, solid and/or cystic tumor, often replacing normal brain tissue (Fig. 1) and sometimes eroding the skull [16]. Most fetuses with prenatally diagnosed intracranial teratomas die before or shortly after birth. The prognosis worsens with increasing tumor size and decreasing gestational age at diagnosis [16-19].

Ultrasonography (US) findings of fetal brain teratoma in a 21-week fetus.
A. A huge solid and cystic mass (arrows) in the posterior cranial fossa is replacing and compressing the cerebral hemispheres (asterisk). B. Color Doppler US shows prominent blood flows in the mass.
Most fetal astrocytomas occur at the supratentorial posteriorcranial fossa. Fetal astrocytomas range from benign to malignant tumors [15]. Glioblastoma accounts for 2%-9% of congenital brain tumors [10] and 60% of fetal brain astrocytoma [15]. On the prenatal US, glioblastoma appears as a large homogeneous hyperechoic mass involving the supratentorial cerebral parenchyma [20]. Mass effect and hydrocephalus are usually accompanied. The prevalence of hemorrhage in congenital glioblastoma has been reported as 18%, which is much higher than in children and adults [10]. The rapid growth of congenital glioblastoma may be related to this higher incidence of bleeding, which explains the poor prognosis of glioblastomas. In a few reported cases, the cause of immediate death was heart failure due to anemia resulting from the sequestration of a large amount of blood by the highly vascular tumor [21].
Fetal intracranial hemorrhage usually mimics brain tumors (Fig. 2). In many instances, the differential diagnosis between intracranial tumor and hematoma is impossible, but several findings may be helpful in the differential diagnosis. Hematoma is usually welldemarcated and shows decrease in size and change in echo pattern at the follow-up US exams.
Ultrasonography (US) findings of intracranial hematoma in a 29-week fetus.
A. The axial US image shows a large mixed echogenic mass (arrow) in the left side of cerebral hemisphere. The falx cerebri (arrowhead) is shifted to right. These findings are very similar to brain tumor, but the margin of the mass is much better defined. B. The coronal power Doppler US image demonstrates no blood flow in the mass.
Tumors of the Fetal Face and Neck
Fetal tumors of the face and neck are a diverse group of lesions.
Vascular lesions including lymphangioma and hemangioma are common, but these two lesions may be considered as congenital malformations rather than true neoplasm [22]. Teratoma of face and neck is relatively uncommon compared with lymphangioma, but is a true neoplasm. The prognosis of teratoma usually depends on the size and location of the tumor related with airway obstruction.
There are four histological types of lymphangioma including capillary lymphangioma, cavernous lymphangioma, cystic hygroma, and vasculolymphatic malformation [23]. These types are considered to be a spectrum of a same pathologic process. Most lymphangiomas occur in the neck, generally in the posterior triangle. US may demonstrate unicameral or multilocular cystic masses with thin or thick-walled septa (Fig. 3). Scattered low-level echoes, a solid component, or fluid-fluid levels, all due to bleeding and fibrin deposition may occur [24]. The internal border of a lymphangioma is usually indistinct and diffuse infiltration may occur [25]. Cystic hygroma usually appears as an isolated malformation and the remainder of the lymphatic system is normal. However, a fetal nuchal cystic hygroma has a poor prognosis when associated with chromosomal abnormalities and fetal hydrops.
Ultrasonography (US) findings of fetal lymphangiomas in a 27-week fetus.
A. The coronal US image of the other fetus shows a multi-septated cystic mass (arrow) in the left side of fetal neck. B. The axial Color Doppler US image reveals no blood flow in the mass (arrow).
Teratomas of face and neck may originate from the thyrocervical area, palate, or nasopharynx, and often appear as bulky masses containing both cystic and solid elements. Calcifications are present in only 50% of cases and may not be obvious at US [26]. Cervical teratomas often involve the thyroid gland, although they do not arise directly from thyroid tissue [4]. Cervical teratomas are usually large at the first detection and may extend to all directions. They may cause marked hyperextension of the fetal neck, resulting in dystocia. Fetal oropharyngeal teratoma is also known as epignathus and is easily detected by prenatal US [27]. It usually appears as a large solid and cystic mass the anterolateral aspect of the fetal face and neck. A stalk of mass is attached to the palate through oral cavity (Fig. 4). Intracranial extension of the mass is not uncommon and the prognosis is not good in such cases. Because of the difficulty in swallowing of amniotic fluid, one-third of these tumors are associated with polyhydramnios [28]. Neonatal survival depends on the size and extent of the involved tissues, respiratory compromise being the major cause of morbidity and mortality. Untreated cervical teratomas are associated with a high mortality rate, though after surgical removal the chances of survival are excellent. There have been several reports [29-31] that the ex utero intrapartum treatment (EXIT) procedure has increased survival rates of fetuses with obstructing neck mass to 69%-92%. In the EXIT procedure, the fetus is partially delivered by cesarean section and undergoes intubation or surgical procedures while the placenta and umbilical cord remain intact.
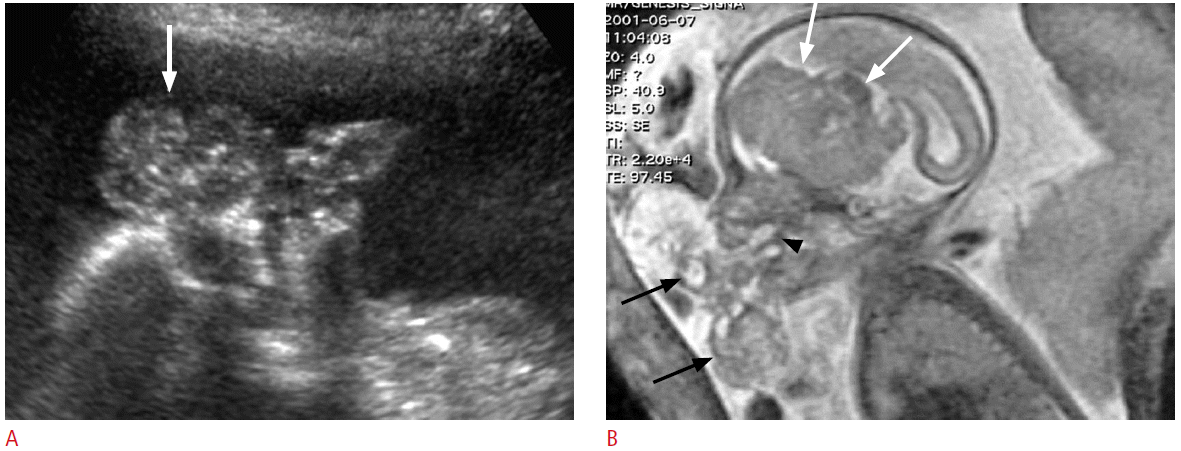
Prenatal ultrasonography (US) and magnetic resonance imaging (MRI) findings of fetal oropharyngeal teratoma in a 32-week fetus.
A. The sagittal US image shows a lobulated solid and cystic mass (arrow) with tiny calcifications in front of the fetal face. B. Fetal MRI reveals a large solid and cystic mass (black arrows) in front of the fetal face and a stalk (arrowhead) abutting to the palate. The mass also shows marked intracranial extension (white arrows).
Tumors of the Fetal Chest
Tumors in fetal chest are very rare condition, but various tumors can develop in fetal chest and heart. Lymphangioma and hemangioma may also be found in the axillae and mediastinum. The US findings are not different to those of cervical tumors.
Congenital infantile fibrosarcoma is a very rare mesenchymal tumor that is primarily developed in the soft tissue of distal extremities and occasionally in unusual locations such as the lung and heart [32,33]. Prenatal diagnosis may be possible from 26 weeks of gestation by US and MRI, but congenital infantile fibrosarcoma is usually diagnosed as hemangioma.
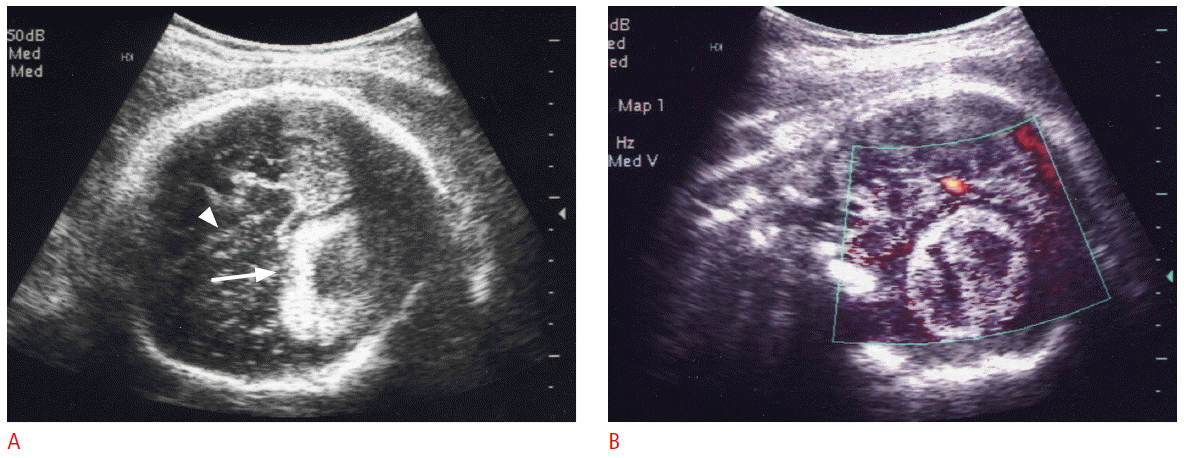
Congenital cardiac tumors are rare, with a prevalence of 0.01%-0.05% [34]. Rhabdomyoma is the most common cardiac tumor in infants and children, accounting for 60% to 86% [34,35] followed by fibroma, teratoma, myxoma, and hemangioma. However, differential diagnosis between these tumors is difficult. Rhabdomyoma may occur as a single or multiple lesions within the ventricles and may be detected prenatally. More than 50% is associated with tuberous sclerosis [36]. The majority of rhabdomyomas are discovered incidentally at routine obstetrical US, whereas a few are discovered in patients with a family history of tuberous sclerosis or fetal cardiac arrhythmia, or in association with nonimmune hydrops. On the prenatal US, rhabdomyoma appears as a homogeneous, echogenic, intracardiac mass, and is most commonly located in the ventricle, especially at the ventricular septum (Fig. 5). Cardiac rhabdomyomas are often multiple, occurring in more than one cardiac chamber and may produce arrhythmias, obstruct blood flow in the heart, or diminish contractility [37]. Although cardiac tumors can easily be detected by prenatal US at as early as 20 weeks of gestation, diagnosis requires that they are of sufficient size. During the fetal period cardiac rhabdomyomas are known to either become larger or regress, but most regress postnatally, either partially or completely [37]. Fetal tuberous sclerosis is usually diagnosed with multiple cardiac rhabdomyomas, but prenatal detection of subependymal hamartoma (Fig. 6) is not easy in many cases. Renal cysts and angiomyolipomas are detected after birth in most of cases.

Ultrasonography (US) findings of cardiac rhbdomyomas in a 34-week fetus with tuberous sclerosis.
A. Four chamber view of fetal heart shows echogenic masses at the interventricular sepum (arrow) and in the left and right ventricles (arrowheads). B. The coronal US image of fetal brain reveals lower echoic hamartomas at the frontal subependymal areas of bilateral lateral ventricles (arrows).
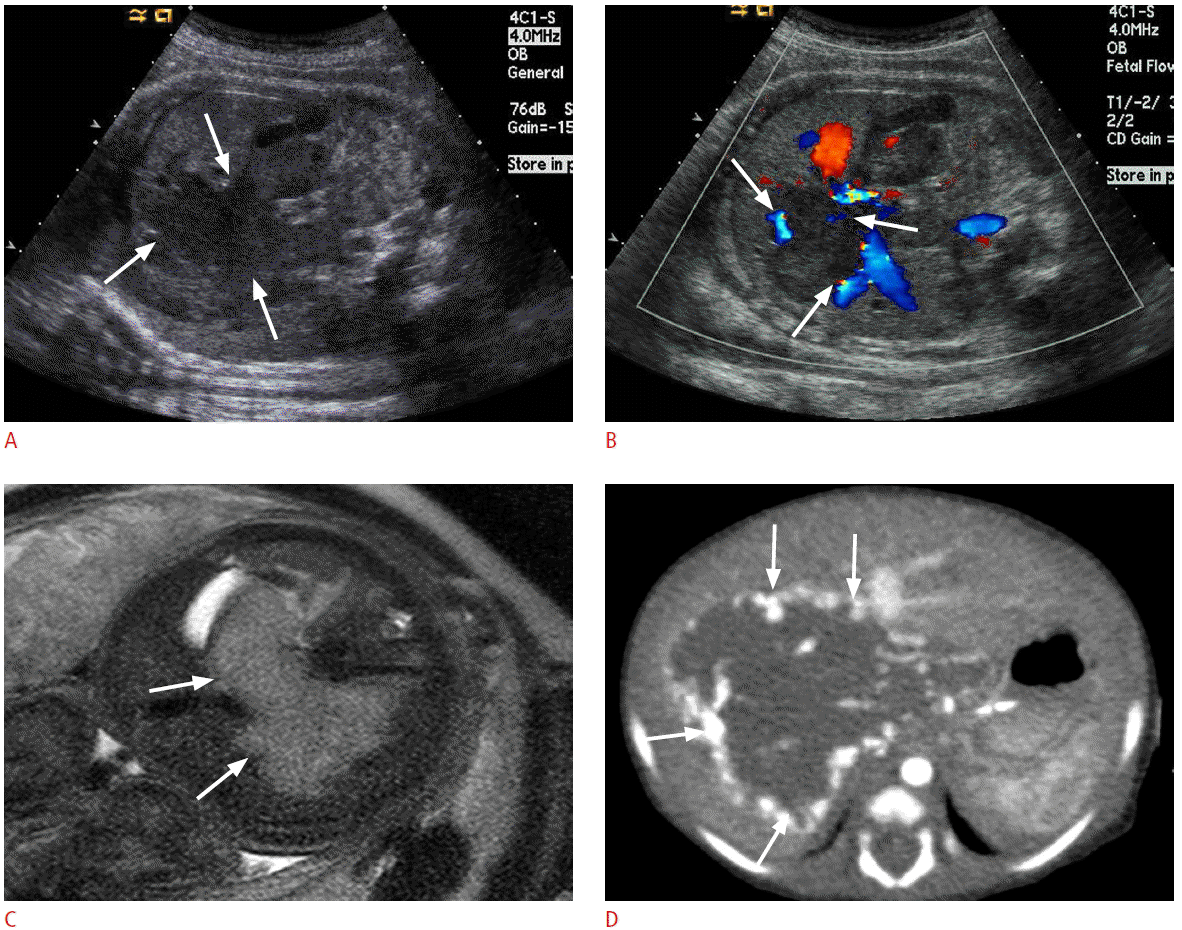
Prenatal and postnatal imaging features of hepatic hemangioendothelioma in a 36-week fetus.
A. Coronal ultrasonography (US) image shows a low-echogenic large mass (arrows) with lobulated contour in the fetal liver. B. Color Doppler US image shows prominent blood flows (arrows) at the periphery of the mass. C. T2 weighted magnetic resonance image reveals a large mass (arrows) with high signal intensity in the fetal liver. D. Postnatal contrast-enhanced computed tomography taken at arterial phase reveals a large liver mass with peripheral bright enhancement (arrows).
Tumors of the Fetal Liver
Liver tumors represent approximately 5% of all congenital tumors [4]. A wide variety of both benign and malignant neoplasms can develop in the fetal liver including hemangioendothelioma, mesenchymal hamartoma, hepatoblastoma, hepatocellular adenoma, and metastases [4]. The imaging findings of fetal hepatic tumors may widely overlap, the early detection and prenatal follow up of these tumors are very important for fetal, maternal, and postnatal care.
The histogenesis of benign vascular liver tumors is not completely clear, which has led to some confusion in terminology. In the literature, vascular tumors are variably described as hemangioma, infantile hemangioma, hepatic arteriovenous malformation, and infantile hemangioendothelioma [22]. Although hemangioma is the most common benign hepatic tumor, prenatal diagnosis liver hemagioma has rarely been reported [38,39]. The true incidence of fetal hepatic hemangioma was certainly underestimated because this tumor is often uncomplicated and resolves spontaneously during the first two years of life. On prenatal US, hepatic hemangioma usually appears as a well-circumscribed abdominal mass with ranging in diameter from 1 to 10 cm. It may be solitary or multiple. Multiple liver hemangiomas sometimes appear as part of generalized hemangiomatosis syndrome. The internal structure is generally heterogeneous, with hypoechoic areas located at center, and both hypo- and hyperechogenicity may be observed. CDUS may show vascular flow with low resistive index (Fig. 6) [38,39]. The mortality rate in infantile hepatic hemangioma has been reported as 12%-90%. Complications include congestive cardiac failure, thrombocytopenia, and intra-abdominal hemorrhage as a consequence of rupture of the hemangioma [40].
Mesenchymal hamartoma is a benign liver tumor composed of large, fluid-filled cysts surrounded by loose mesenchymal tissue containing small bile ducts [41]. It usually appears as a predominantly cystic or mixed echogenic mass and should be a primary consideration in the differential diagnosis of an enlarging cystic liver lesion [22]. Prenatal US finding of mesenchymal hamartoma widely overlaps with that of hepatic hemangioma. Usually it does not have increased vascularity at CDUS [42,43].
Hepatoblastoma is the most common congenital hepatic malignancy, but it is less common during fetal life than hemangioma or mesenchymal hamartoma [4]. Hepatoblastoma usually appear as a well-defined echogenic lesion, which may have a spoke-wheel appearance [44]. Calcifications and pseudo-capsule may be common features of hepatoblastoma [4]. The prognosis of perinatal hemangioma is very poor and complications, including intrapartum rupture, hemorrhage, hydrops, and metastases, are common [4].
Hepatocellular adenoma in the fetus is very rare and its radiological findings have not been extensively described. These tumors are generally round masses with variable echogenicity, and are clearly distinguishable from normal liver parenchyma. They are usually quite large, and infarction, hemorrhage and rupture are frequent. At postnatal computed tomography and MRI, internal hemorrhage, fatty change, or necrosis may be observed.
Tumors of the Fetal Kidney and Adrenal Gland
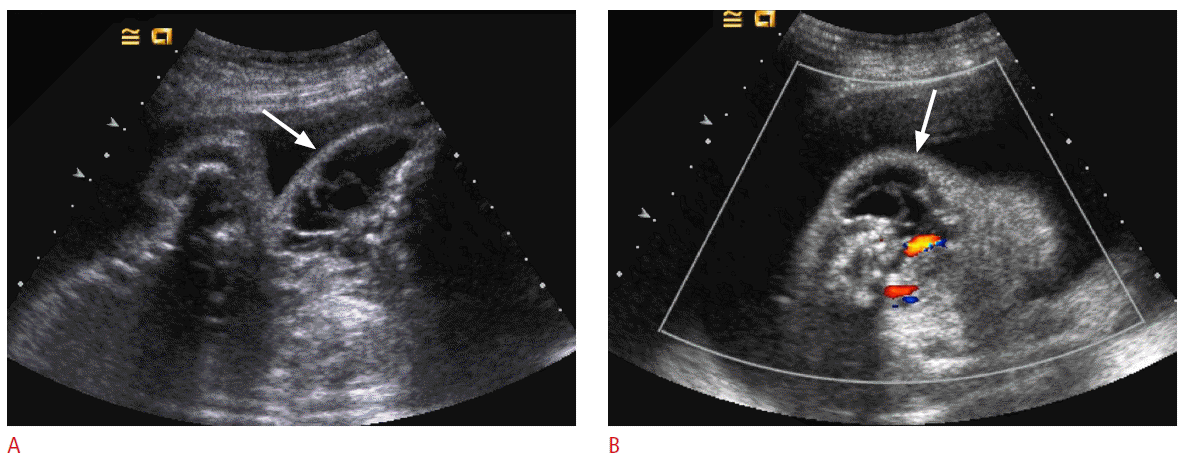
About 5% of perinatal tumors develop from the kidney. The most of perinatal renal tumor is mesoblastic nephroma. Fetal or perinatal Wilms tumor and rhabdoid tumor are very rarely reported [4]. Mesoblastic nephroma is a rare benign mesenchymal renal tumor, arising either from the kidney or the renal fossa. Fetal mesoblastic nephroma occurs exclusively during the third trimester. Mesoblastic nephromas tend to be large, involving at least half of the kidney, and are well circumscribed although not encapsulated (Fig. 7). Rapid enlargement of the mass may lead to hemorrhage. Mesoblastic nephroma is frequently associated with polyhydramnios and consequently with premature labor. The cause of the polyhydramnios is not fully understood [4,22]. Another associated abnormality is hypercalcemia [4,45], which may cause polyuria and polyhydramnios [43]. The prognosis after surgical excision is usually good. On prenatal US, mesoblastic nephroma usually appears as a well-defined large homogeneous mass with preserved contour of involved kidney. CDUS usually shows increased blood flow, which may lead to fetal hydrops in some cases [4,46].
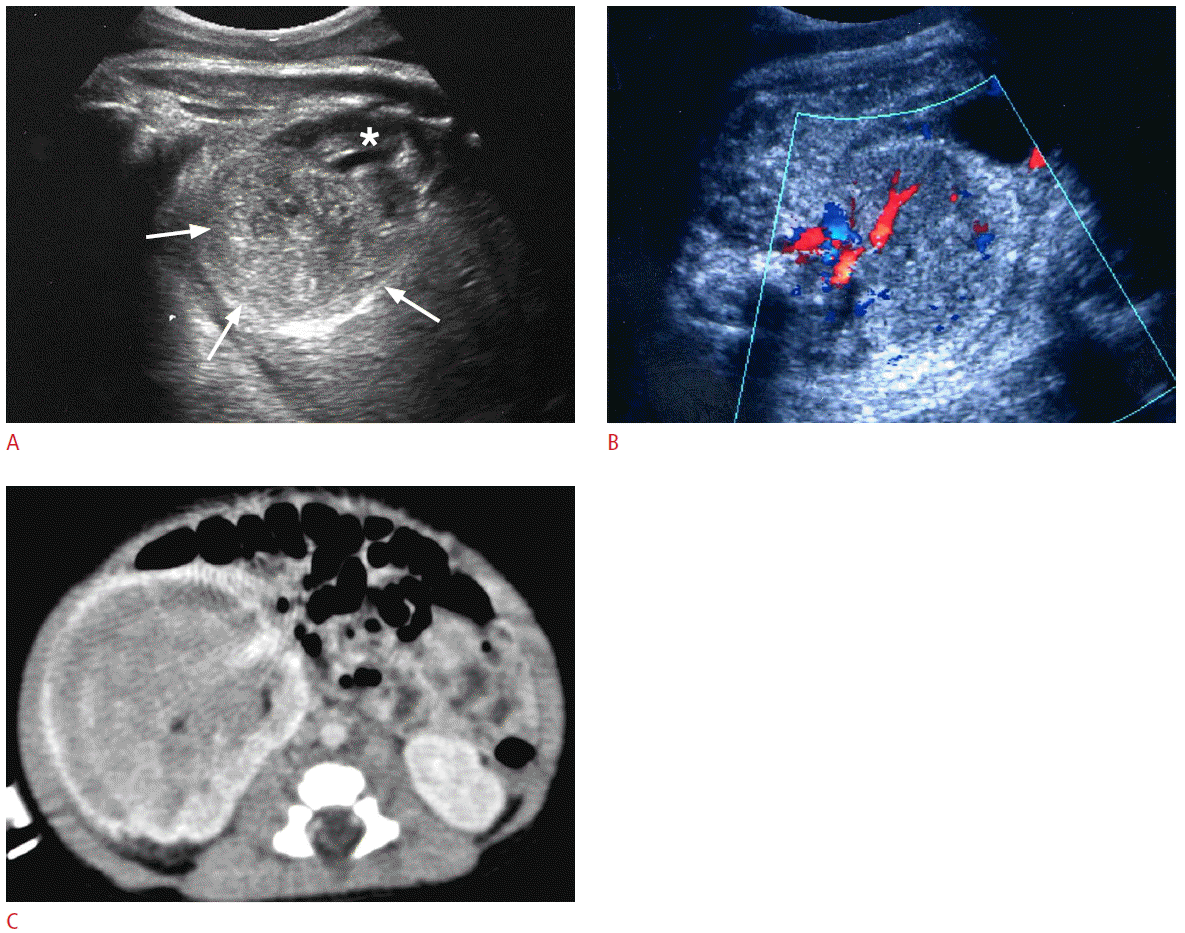
Prenatal and postnatal imaging findings of mesoblastic nephroma in a 35-week fetus.
A. A large heterogeneous mass (arrows) is replacing the lower two third of the right kidney (asterisk). B. Color Doppler ultrasonography reveals prominent blood flows in the mass. C. Contrast-enhanced computed tomography taken one week after birth reveals a huge mass replacing almost portion of the right kidney.
Neuroblastoma is the most common malignant tumor in neonates, accounting for 20% of all such malignancies [47]. More than 90% of fetal neuroblastomas develop in the adrenal gland, while only 35% of pediatric cases occur in adrenal gland [48,49]. Fetal neuroblastomas are usually detected in the third trimester of pregnancy and are typically suprarenal and right sided [50,51]. The US findings are variable, ranging from pure cystic to solid and small to large masses (Fig. 8). Small fetal neuroblastoma usually appears as a hyperechoic mass [49,51,52], which should be distinguished from extralobar pulmonary sequestration (Fig. 9) or perirenal fat tissue [50]. Overall prognosis of fetal neuroblastoma is excellent, and this may be due in part to the detection of an otherwise clinically silent neuroblastoma in situ, which would undergo spontaneous regression [53]. It may be appropriate to avoid overly aggressive therapeutic interventions, especially when many favorable prognostic indicators are present at diagnosis.
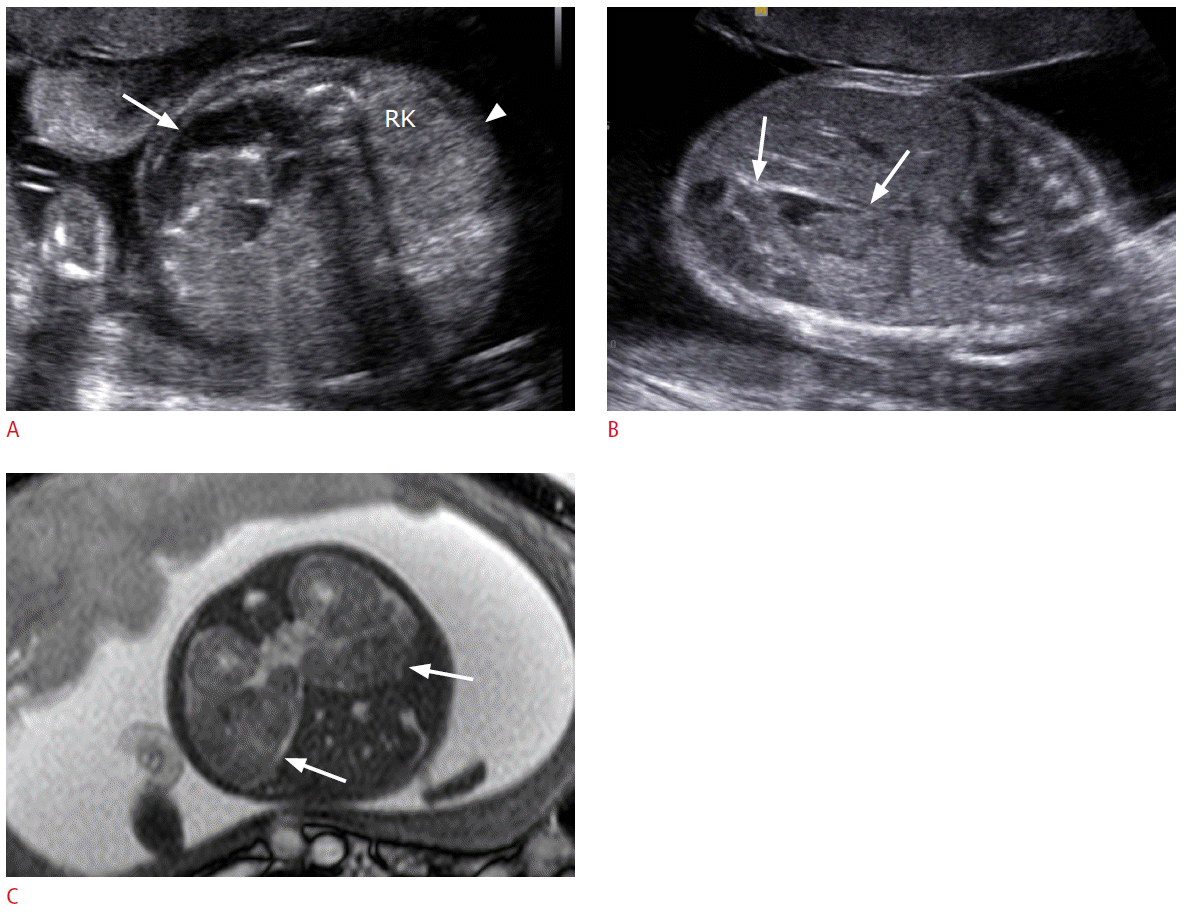
Prenatal ultrasonography (US) and magnetic resonance imaging (MRI) findings of neuroblastomas in a 27-week fetus.
A. The axial image of fetal abdomen shows a large heterogeneously echogenic mass (arrow) in the left posterior abdomen and a small homogeneously hyperechoic mass (arrowhead) anterior to the right kidney (RK). B. The sagittal US image shows a large heterogeneous mass (arrows) in left posterior abdomen of the fetus. C. Fetal MRI reveals two masses (arrows) in bilateral anterior perirenal spaces.
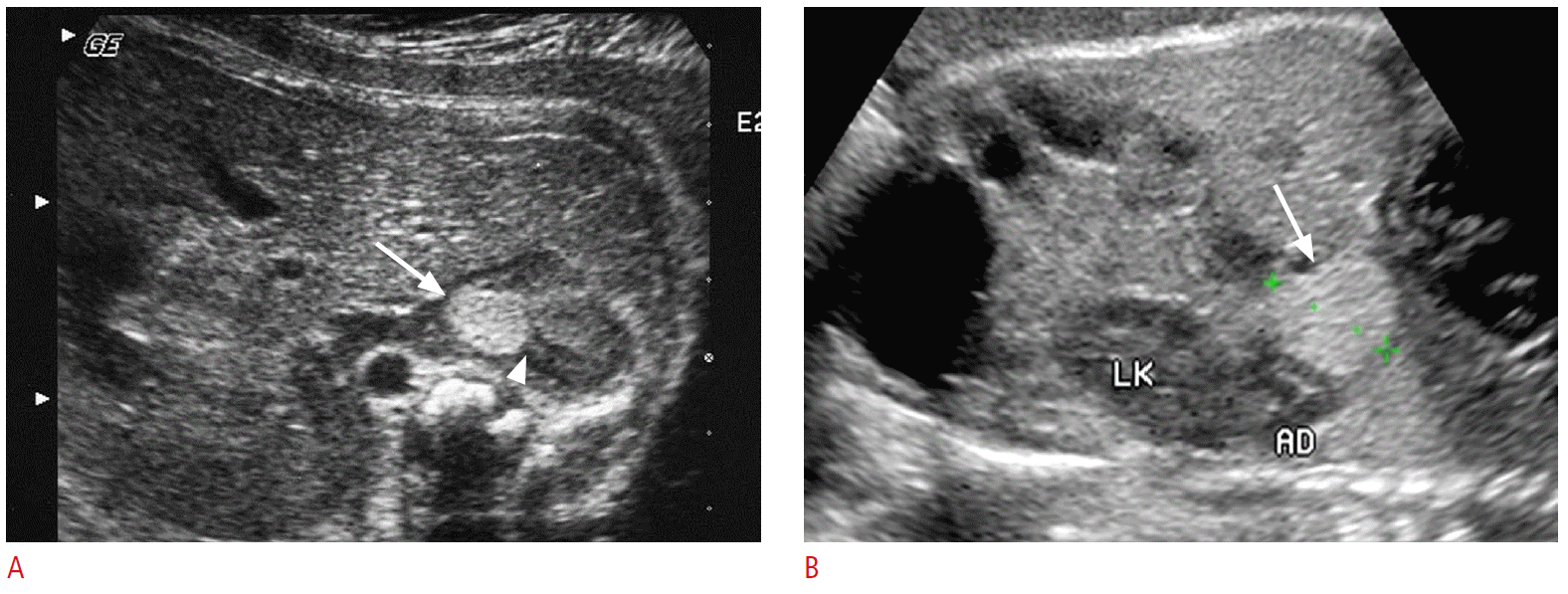
Comparison of prenatal ultrasonography (US) findings of small neuroblastoma (A) and extralobar pulmonary sequestration (B).
A. The axial US image of a 32-week fetus shows a round echogenic mass (arrow) at the right adrenal gland. A part of adrenal gland is obliterated by the mass (arrowhead). B. The coronal US images of a 32-week fetus show an echogenic mass (arrow) at the left subdiaphragmatic area, mimicking neuroblastoma. Abutting left adrenal gland (AD) and kidney (LK) are intact.
Tumors of the Fetal Pelvic Cavity
Sacrococcygeal teratoma is the most common congenital neoplasm. The prevalence was reported as one in 40,000 live births with a female preponderance of 4:1 [4]. The fetal prevalence is most likely higher, considering the high number of in utero deaths, stillbirths, or terminated pregnancies that may go underreported [22]. It usually appears as a large midaxial exophytic mass at the perineum. It may be almost entirely external (type I), internal and external in equal parts (type II), mainly internal (type III), or entirely internal (type IV) [54]. At prenatal US, the majority of those tumors are seen as a solid, or mixed cystic and solid, external caudal mass (Figs. 10, 11). Only small proportions are entirely cystic [55]. A sacrococcygeal teratoma may cause significant perinatal morbidity and mortality, and may lead to various complications such as severe dystocia, fetal hydrops, polyhydramnios, or bleeding during parturition. Due to vascular steal from high metabolic demand and secondary highoutput cardiac insufficiency, the prognosis for fetuses with solid tumor is usually poor [56]. The mirror syndrome is a life-threatening condition with maternal fluid retention and hemodilution mimicking severe preeclampsia [57], which occurs in the setting of hydrops fetalis and manifests as progressive maternal edema “mirroring” that of the sick fetus.

Prenatal ultrasonography (US) findings of a large sacrococcygeal teratoma in a 21-week fetus.
A. The sagittal US image shows a large mainly cystic protruding mass (arrow) at the perineum of the fetus. B. Color Doppler US reveals subtle blood flows at the solid portion of the mass.
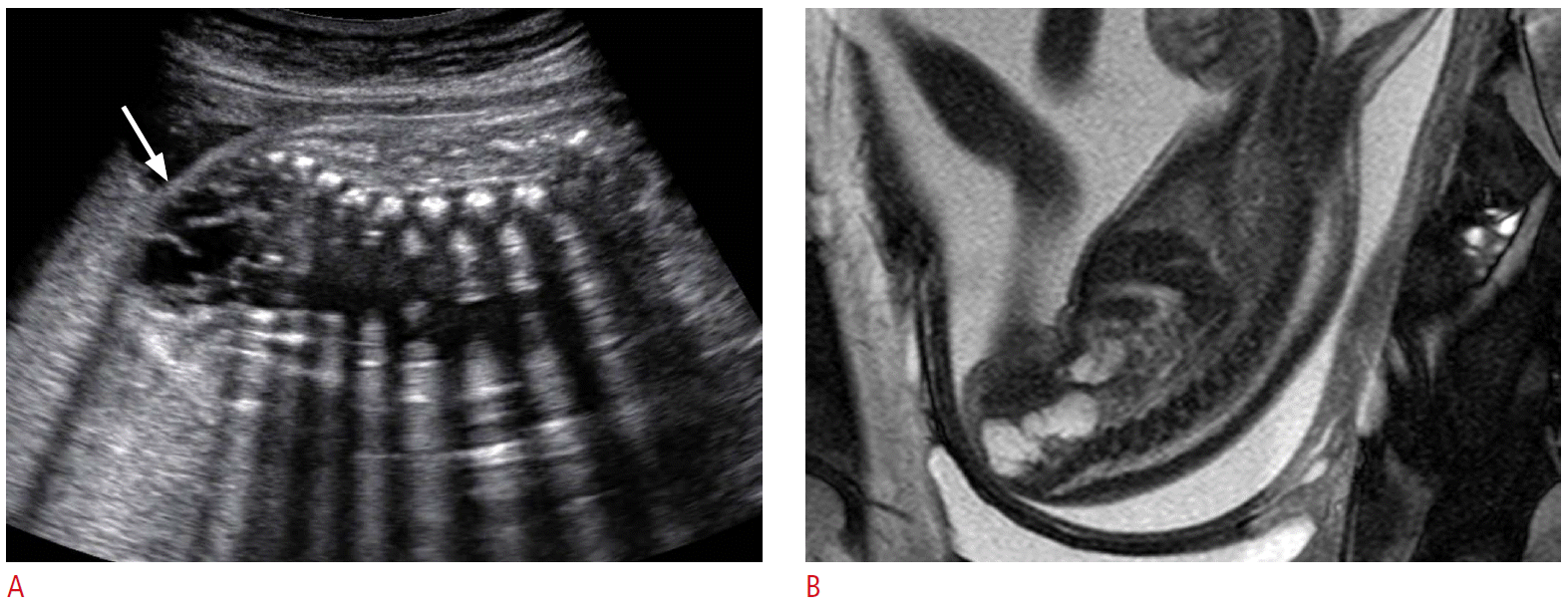
Prenatal ultrasonography (US) and magnetic resonance imaging (MRI) findings of mainly intrapelvic sacrococcygeal teratoma in a 24-week fetus.
A. A lobulated contour multi-septated cystic mass (arrow) at the perineum of the fetus. B. Sagittal T2-wighted magnetic resonance image reveals a multi-septated cystic mass at the anterior portion of the sacrum and coccyx.
Rhabdomyosarcoma is the most common soft-tissue tumor in children, but very rare in fetuses. The most common locations are the head and neck (41%), the genitourinary tract and the abdominal cavity (34%) and the trunk or extremities (25%) [58]. Prenatal US may reveals a large echogenic soft-tissue mass replacing the normal tissues (Fig. 12) [59]. Differential diagnosis from other soft tissue tumors, such as teratoma and fibrosarcoma, is usually impossible and postnatal biopsy is necessary. The prognosis depends on the stage of the disease, age, the primary site, and subtype of the tumor. The embryonal subtype is most common in children and has the best prognosis, but all have a poor prognosis if they occur in perinatal period and are in an advanced stage [59,60].
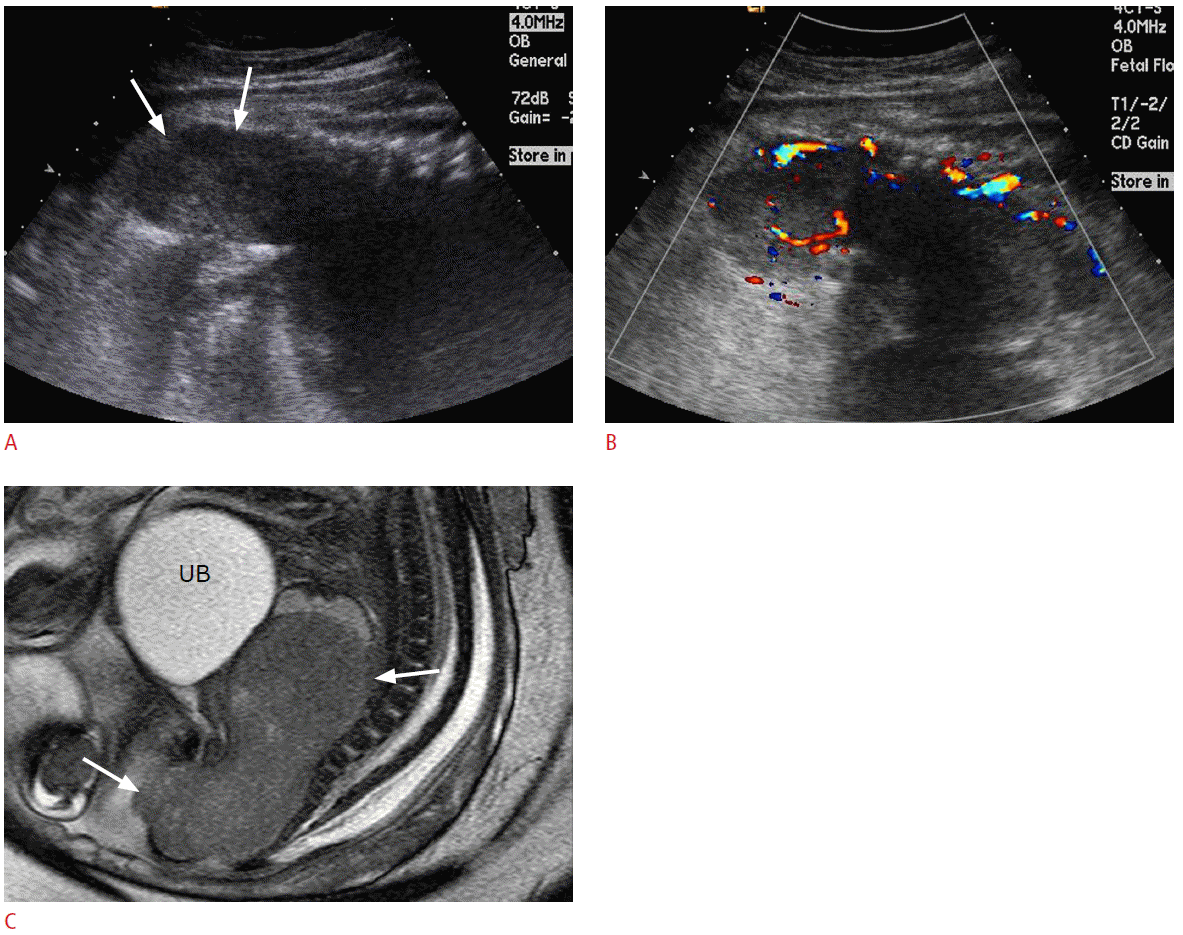
Prenatal ultrasonography (US) and magnetic resonance imaging (MRI) findings of rhabdomyosarcoma at the pelvis of a 36-week fetus.
A. A sagittal US image of fetal pelvis shows a large mass (arrows) in front of the sacrum and coccyx mimicking intrapelvic solid teratoma. B. Color Doppler US shows prominent vascular flows in the mass. C. T2-weighted magnetic resonance image reveals a well-demarcated solid mass (arrows) at the posterior portion of the lower pelvic cavity. UB, urinary bladder.
Conclusion
Although tumors rarely occur during the fetal life, variety of tumors can be detected in each fetal organ. Fetal tumors are usually different in their histologic characteristics, anatomic distribution, pathophysiology, and biologic behavior compared with those of the same tumors in children. The routine antenatal US exams have increased detection and diagnosis of fetal tumors and MRI may be helpful for the differential diagnosis in some cases. Although several fetal tumors may mimic other common fetal anomalies, some specific imaging features may carry early accurate diagnosis of fetal tumors. It is very important for the diagnostic physician to be familiar with imaging features and pathophysiological characteristics of fetal tumors, which may alter the prenatal management of a pregnancy and the mode of delivery, and facilitate immediate postnatal treatment.
Notes
No potential conflict of interest relevant to this article was reported.
