Introduction
Piriformis syndrome is attributed to sciatic nerve compression caused by hypertrophy, inflammation, or anatomical deformities of the piriformis muscle. The sciatic nerve is the largest nerve in the body. It runs under the piriformis muscle, passes through the greater sciatic foramen, and then passes between the greater trochanter and ischial tuberosity along the back of the thigh [1]. The possible anatomical relationships between the piriformis muscle and sciatic nerve are classified into six types: an undivided sciatic nerve passing below the piriformis muscle, a divided sciatic nerve passing through and below the piriformis muscle, a divided nerve passing above and below the muscle, an undivided sciatic nerve passing through the piriformis muscle, a divided nerve passing through and above the muscle, and an undivided sciatic nerve passing above the piriformis muscle [2].
Common symptoms of piriformis syndrome include pain in the buttock or thigh and sciatica, in which the intensity of pain may increase when the patient is seated for a long period. Approximately 6%-8% of cases of lower back pain accompanied by sciatica have been reported to be related to this syndrome [3]. Since piriformis syndrome does not have any distinguishing clinical symptoms or specific diagnostic criteria, it is difficult to diagnose and is frequently misdiagnosed [4]. Conservative approaches can be used for treatment. These include relaxing the tense piriformis muscle, reducing sciatic nerve compression through stretching exercises, massage, or thermotherapy, and administration of non-steroidal anti-inflammatory drugs or muscle relaxants. However, when these methods are not effective, direct injection of local anesthetics, steroids, or botulinum toxin around the piriformis muscle can decrease hypertrophy or inflammation. Previously, injections were administered blindly in the area of severe pain or after confirming the location of the piriformis muscle using electromyography, computed tomography, or magnetic resonance imaging [4-6].
In the meantime, ultrasound (US)-guided injection techniques have been reported in recent studies [7-11]. This technique is more convenient with no radiation hazard. Several studies have evaluated the efficacy of US-guided techniques; however, none followed up on patients for longer than 3 months [11]. The purpose of this study was to retrospectively analyze the efficacy of US-guided injections in patients with piriformis syndrome for a maximum of 3 years based on follow-up observations in their medical records.
Materials and Methods
This study was approved by the Institutional Review Board of our hospital. A waiver of informed consent was obtained for this retrospective study. The subjects for this study were chosen from patients diagnosed in a single tertiary hospital, from January 2010 to October 2012. Based on patient history and the results of electromyography and flexion-adduction-internal rotation tests, 68 patients were clinically diagnosed with piriformis syndrome and referred to the radiology department for injection therapy. A single radiologist retrospectively analyzed the medical records of the subjects. Five patients were excluded: one patient for whom follow-up records were unavailable and four patients who responded to the epidural steroid injection but not to the piriformis muscle injection. The study ultimately included 23 men and 40 women, with an average age of 63.2 years (±14.3 years; range, 24 to 90 years). The follow-up time after the US-guided steroid injection varied from 1 to 36 months (mean, 15.1 months). Among the 63 patients, 26 received an additional spinal epidural steroid injection between 2 days and 2.25 years after the initial injection. The possibility of spinal disease in these patients could not be ruled out. The patients were divided into two groups. The first group (37 subjects) received a US-guided steroid injection only around the piriformis muscle. The second group (26 subjects) received both piriformis muscle and spinal epidural injections. The first group included 14 men and 23 women, with an average age of 60.6 years (±0.6 years; range, 28 to 90 years). The second group included 10 men and 16 women, with an average age of 66.9 years (±6.9 years; range, 24 to 87 years).
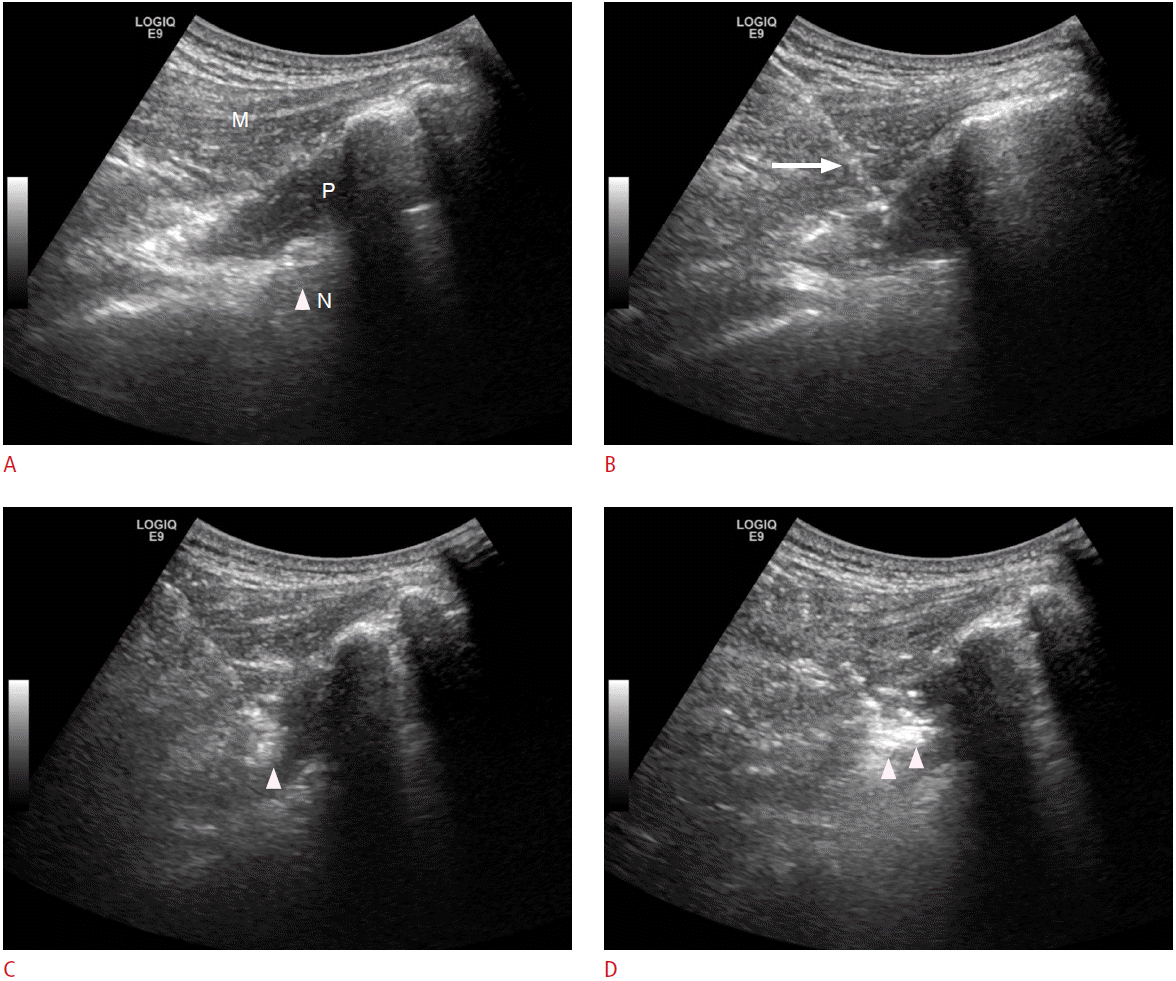
US-guided steroid injections were administered by one of two musculoskeletal radiologists with 7 and 14 years of experience, respectively. After placing the patients in a prone position on the bed, a curvilinear US probe was positioned at the sacral hiatus for a transverse scan. It was drawn towards the greater trochanter in an outward direction and above the level of the sacrospinous ligament to identify the piriformis muscle and the sciatic nerve. These structures are located deep in the caudal muscle belly of the gluteus maximus muscle, between the outer border of the sacral vertebrae and inner border of the greater trochanter. Forty milligrams of triamcinolone acetonide (40 mg/mL, Triam, Shin Poong Pharm, Korea) was then injected around the piriformis muscle, using a 22-gauge spine needle under US guidance (Fig. 1).
The efficacy of the injections was assessed using the visual analog scale score recorded in the medical record at each hospital visit at the commencement of treatment and throughout the follow-up period. Improvement was defined as pain reduction of 50% or more compared to the initial pain level, as assessed by the visual analog scale. Partial improvement was defined as less than a 50% reduction in pain compared to the initial level. Failure was defined as no pain reduction or worsening of pain.
If the pain worsened during the follow-up visits, which spanned 3 years, the patient was said to have had a relapse even if improvement or partial improvement had been noted during the follow-up visit that took place 1-month postinjection. The response of patients who received repeated injections was also assessed using the visual analog scale.
The mean visual analog scores before and after steroid injections were analyzed separately in the two groups of patients. The paired t test was used to compare the mean values within each group. The number of patients who were treated once or twice was tabulated for each treatment group. All statistical tests were performed using SPSS ver. 18 (SPSS Inc., Chicago, IL, USA), and P-values of <0.05 were considered to indicate statistical significance.
Results
Of the 63 subjects, 33 (52.4%) patients were observed for less than 1 year. The number of patients was 11 (17.5%) and 19 (30.1%) at the 2-year and 3-year follow-ups, respectively (1.79±0.90 years; range, 1 to 3 years). In the first group, 15 patients (40.5%) showed improvement, seven (18.9%) showed partial improvement, and 15 (40.5%) failed to respond to the initial treatment. In the second group, eight patients (30.8%) showed improvement, 11 (42.3%) showed partial improvement, and seven (26.9%) failed to respond to the initial treatment (Tables 1, 2).
Of the 18 patients from both groups who showed partial improvement, four underwent a subsequent piriformis injection. The average time that elapsed between the first and second piriformis injections was 161 days (range, 18 to 482 days). Based on the 3-year follow-up records of four patients who underwent a repeat injection, two showed improvement and two experienced no effect. There was no case of relapse in this group. There was no recurrence during the follow-up period of 1 to 36 months. After the second injection, one female patient (1.6%) reported facial flushing for 4 days starting from the day of the procedure; however, the symptom resolved spontaneously. No other side effects were observed among the other 62 patients.
Therefore, considering the effects of the initial treatment along with any subsequent treatments, 25 patients (39.7%) showed improvement, 16 (25.4%) showed partial improvement, and 22 (34.9%) failed to respond to the treatment.
Discussion
The term piriformis syndrome was first used in 1947 by Robinson [12], who described six characteristics. These include a history of trauma or direct fall on the hips; the presence of pain in the hips or the sacroiliac joint that occasionally causes a gait abnormality; hip atrophy; palpable sausage-shaped lumps; a positive Lasegue sign (in sciatica, pain from bending at the hips with the knees in extended position); and aggravation of pain when lifting or bending the legs [12].
Piriformis syndrome is difficult to diagnose in patients with lower back pain and symptoms of sciatica, and only about 6%-8% of such patients are actually diagnosed with piriformis syndrome [3]. After spinal disease is excluded as a cause of lower back pain and symptoms of sciatica, piriformis syndrome is one of the diseases that should be suspected. Piriformis syndrome patients may show normal results on an electromyogram. According to a previous study, the flexion-adduction-internal rotation test has 85% specificity in identifying piriformis syndrome and 82% specificity in identifying normal legs [13].
Previous studies have reported using US-guided steroid injections as one of the treatment methods for piriformis syndrome [7], and the appropriate clinical technique has also been described [9]. However, no studies have yet addressed the long-term efficacy of US-guided steroid injections. Cassidy et al. [1] performed injection therapy using a mixture of a local anesthetic and a steroid on 468 patients with suspected piriformis syndrome who tested positive on the flexion-adduction-internal rotation test, and 370 of these patients (79%) reported at least 50% improvement in symptoms. The average length of observation in their study was 10.2 months. However, their study did not present details regarding the injection methods, including whether the piriformis muscle was injected directly [1]. In a study by Reus et al. [11], US-guided steroid injections of a mixture of mepivacaine and triamcinolone were administered near the sciatic nerve in 10 patients with piriformis syndrome, of whom eight (80%) showed alleviation of symptoms within a week, while the remaining two patients (20%) showed improvement within 2 months [11]. In contrast, in another study that evaluated the efficacy of surgical treatment in 82 patients with piriformis syndrome who were followed up for 2 years, 76 patients (92.7%) showed alleviation of symptoms [14].
A limitation of our study was its retrospective design, which meant that the analysis of post-treatment efficacy was based solely on data from the patients’ medical records. Another limitation was that it is difficult to make an accurate diagnosis of piriformis syndrome, which complicated the selection of subjects. An additional limitation was the relatively small number of patients (n=63) who were evaluated. Future studies aimed at establishing objective criteria for pain alleviation and devising US-based or magnetic resonance imaging-based methods for accurate diagnosis are needed and are currently being planned.
In conclusion, US-guided injection may be an effective, safe, and easy treatment for piriformis syndrome, and this technique may therefore be considered as an option for treating patients with piriformis syndrome.



 Print
Print facebook
facebook twitter
twitter Linkedin
Linkedin google+
google+
 Download Citation
Download Citation PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC




