Introduction
With the exponential development of diagnostic and therapeutic technology, the overall survival of patients who have been diagnosed with a specific neoplasm has increased. Along with prolonged patient survival, detection of secondary involvement of the malignant neoplasm in other organs, that is, distant metastases, has also increased [1,2]. Detection and diagnosis of distant metastases has critical significance, since this directly affects the prognosis and treatment planning.
Theoretically, distant metastasis can involve any organ of the body, including the thyroid gland. Metastasis to the thyroid gland is rarely observed in clinical practice, but overall incidences have been reported to range from 1.9% to 24% in various autopsy series [3-5], with metastasis from the kidney, breast, and lungs most frequently reported [4,6,7]. Asymptomatic thyroid nodules are nowadays commonly found due to widespread ultrasonography (US) examinations, and when considering the clinical significance of metastatic carcinoma to the thyroid in cancer patients, differential diagnosis between primary thyroid disease and metastatic ones is essential. US is a safe, easy, and accurate imaging modality that shows excellent performance when used in imaging of the thyroid and adjacent neck areas, and it enables US-guided fine-needle aspiration (US-FNA), which is considered the gold standard in the preoperative diagnosis of various thyroid lesions [8-10]. Although reported in fragments in studies on the clinical characteristics and prognosis of thyroid metastases [3,4,11,12], to our knowledge only a few studies have reported in general on the US features of metastatic carcinoma of the thyroid [13-15]. The purpose of this study was to evaluate the characteristic US features of metastatic carcinoma of the thyroid, and how accurate these imaging features and cytology results obtained from US-FNA are in the diagnosis of metastases to the thyroid gland.
Materials and Methods
Patients
This retrospective study was approved by our Institutional Review Board (IRB) and neither patient approval nor informed consent was required for the review of medical records or US images. Informed consent was signed and obtained from all patients prior to US-FNA or surgery.
From January 2003 to December 2009, we searched the medical records database of our institution for the diagnosis of ‘thyroid metastases.' During this period, 27 patients were diagnosed with metastatic lesions to the thyroid gland by US-FNA, US-guided core needle biopsy (US-CNB) or surgery. Among them, 3 were excluded for the following reasons: metastasis originating from direct invasion of the primary mass located near the thyroid such as the thymus and esophagus (n=2) or failure to obtain US images (n=1). One case that had been published in a previous report was also excluded [16]. A total of 23 thyroid nodules in 23 patients were included in this study. The medical records, US images and radiological reports, and cytopathologic reports of these patients were retrospectively reviewed.
US Imaging and US-FNA Procedures
US was performed in all patients using a 7- to 15-MHz linear array transducer (HDI 3000 or 5000; Philips Medical Systems, Bothell, WA, USA) or a 5- to 12-MHz linear array transducer (iU22; Philips Medical Systems). Compound imaging was obtained in all images using HDI5000 or iU22 machines.
Real-time US was performed by 1 of 5 board-certified radiologists with 1-15 years of experience in thyroid imaging. To reduce the interobserver variability of the US features of the thyroid masses, the US images were retrospectively reviewed by 1 radiologist (JHY) with 4 years of experience in thyroid imaging. This radiologist was informed of the presence of metastatic carcinoma to the thyroid in the patients during image review. The general US appearance of the lesions diagnosed as thyroid metastasis was categorized as follows: mass-forming, for lesions that showed obvious borders between the metastatic lesion and surrounding thyroid parenchyma, and non-mass-forming, for lesions that did not show clear-cut borders between the metastatic lesion and thyroid parenchyma.
The US features of the mass-forming metastatic lesions were classified into the following categories: internal component (solid, mixed solid and cystic, or cystic), echogenicity (hyper- or isoechoic, hypoechoic compared to the thyroid parenchyma, or markedly hypoechoic compared to the strap muscle), margin (circumscribed, microlobulated, or irregular), presence of calcifications, and shape (parallel, defined as being greater in its transverse dimension than the anteroposterior dimension; or non-parallel, defined as being greater in its anteroposterior dimension than the transverse dimension) [17]. Malignant US features were defined as marked hypoechogenicity, non-circumscribed margins, presence of calcifications, and non-parallel shape, based on previously published criteria [17]. Final assessments of the thyroid nodules were given as ‘probably benign' when none of the suspicious US features described above were present or ‘suspicious malignancy' when 1 or more suspicious US features described above were present. Vascularity measured on power Doppler US was available in 16 patients, and was categorized into no, peripheral, or intranodular vascularity [18].
The extent of the metastatic lesion to the thyroid was classified into one of three categories: single; multifocal, defined as multiple lesions showing similar US features to the biopsy-proven index mass involving one thyroid lobe; or bilateral, defined as multiple suspicious masses similar to the index mass involving both thyroid lobes [5]. The cervical areas were thoroughly evaluated during US for the presence of metastatic lymph nodes, and lymph nodes showing 1 or more of the following US features were considered suspicious: loss of fatty hilum, cystic change, round shape (long/transverse diameter ratio <1.5), or mixed and peripheral vascular pattern on power Doppler [19,20].
US-FNA and Cytological Analyses
US-FNA or US-CNB was subsequently performed by the same radiologist who obtained the real-time US images. In cases of patients with multiple lesions, US-FNA or biopsy was performed targeting the lesion with the most suspicious US feature, or at the largest mass when multiple lesions showed similar US features. USFNA was performed with a range of 2 to 4 passes from the targeted thyroid nodule using a 23-gauge needle attached to a 20-mL disposable syringe with an aspirator or a 23-gauge needle attached to a 2-mL disposable syringe without an aspirator, depending on the radiologist's preference. US-CNB was performed in 3 patients for more confirmative pathology results after ambiguous cytology results (n=1), and for additional immunohistochemistry staining for differential diagnosis after being diagnosed with metastasis on cytology (n=2). Using an 18-gauge automated core biopsy needle and a spring-loaded biopsy gun (Promac 2.2L; Manan Medical Products, Northbrook, IL, USA), two cores were routinely obtained from each patient. Local anesthesia was not routinely applied during US-FNA but was used during US-CNB. Aspirated material from USFNA was expelled onto glass slides, smeared, and immediately placed in 95% alcohol for Papanicolaou staining. The remaining material in the syringe was rinsed in normal saline for cell block processing.
One of 5 cytopathologists specializing in thyroid pathology interpreted the slides or specimen obtained from US-FNA or USCNB, respectively. The cytopathologists were not present during biopsy procedures, and additional staining was performed on a case-by-case basis at the suggestion of the cytopathologist.
Results
Clinical Features of the Patients
The clinical features of the 23 patients are summarized in Table 1. Of the 23 patients included in this study, 13 (56.5%) were men and 10 (43.5%) were women. The mean age of the patients at the time of diagnosis of thyroid metastasis was 66.7 years (range, 46 to 85 years). Metastatic lesions to the thyroid were detected by the presence of symptoms or abnormal features during physical examination (n=8), or incidentally by various imaging modalities such as US, computed tomography (CT), or positron emission tomography (PET; n=15).
The locations of the primary tumor in the 23 patients with thyroid metastasis were as follows: lung (n=9), colon (n=3), breast (n=3), head and neck (n=2), uterine cervix (n=1), stomach (n=1), gallbladder (n=1), kidney (n=1), and primary unknown (n=2). Fourteen patients (60.9%) had metastatic lesions in organs other than the thyroid, such as the liver, lungs, bone, and brain. Thyroid metastasis was detected during initial diagnosis and routine workup for cancer staging of the primary tumor in 10 patients. The mean interval between the primary tumor diagnosis and thyroid metastasis of the remaining 13 patients was 62.8 months, ranging from 6 to 192 months.
Two of the patients had lobectomy performed simultaneously to the operation on the primary tumor, of which both were confirmed to have metastatic thyroid carcinoma by surgical pathology. Fifteen patients were treated with chemotherapy, while the remaining 8 patients were referred to hospice care. Twenty-one patients have expired and 2 are alive to the present date, having survived for 46 and 60 months, respectively. The mean survival time of the 21 patients was 9.5 months, ranging from 2 to 38 months. The two patients who are alive have been treated with chemotherapy without surgery.
US Features and US-FNA of Thyroid Metastases
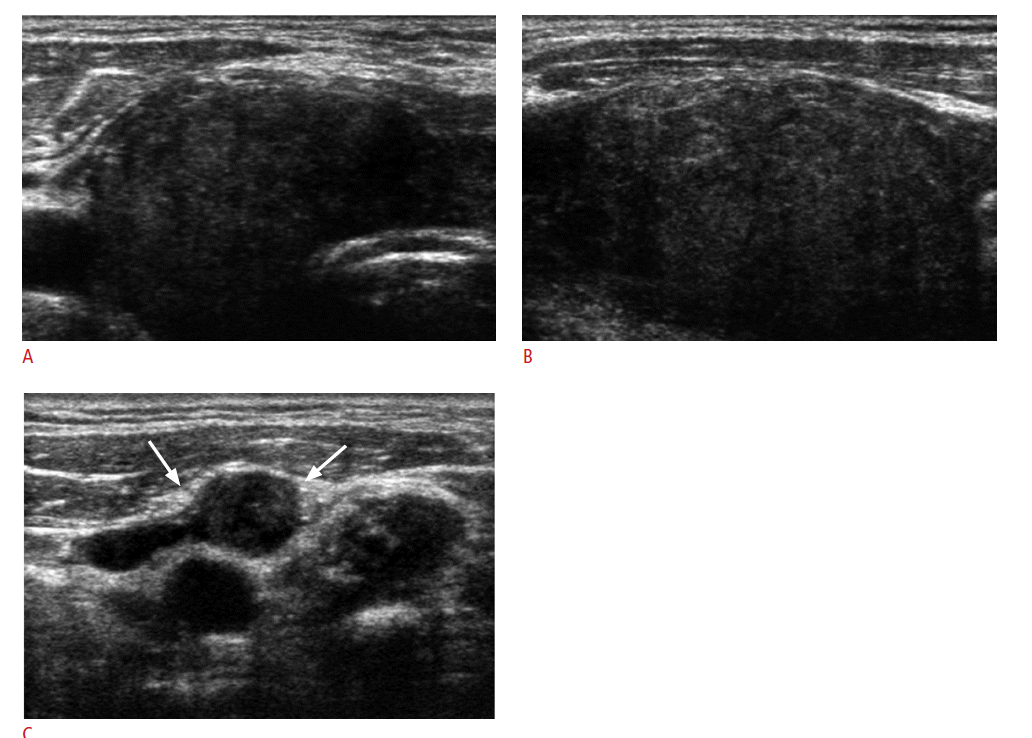
The mean size of the metastatic lesions to the thyroid measured on US was 27.2 mm (range, 5 to 58 mm). The US appearance of the 23 thyroid lesions were mass-forming in 21 (91.3%) and non-massforming in 2 (8.7%) (Fig. 1). Seventeen of the metastatic lesions (73.9%) presented as single lesions, while the remaining 6 (26.1%) showed bilaterality.
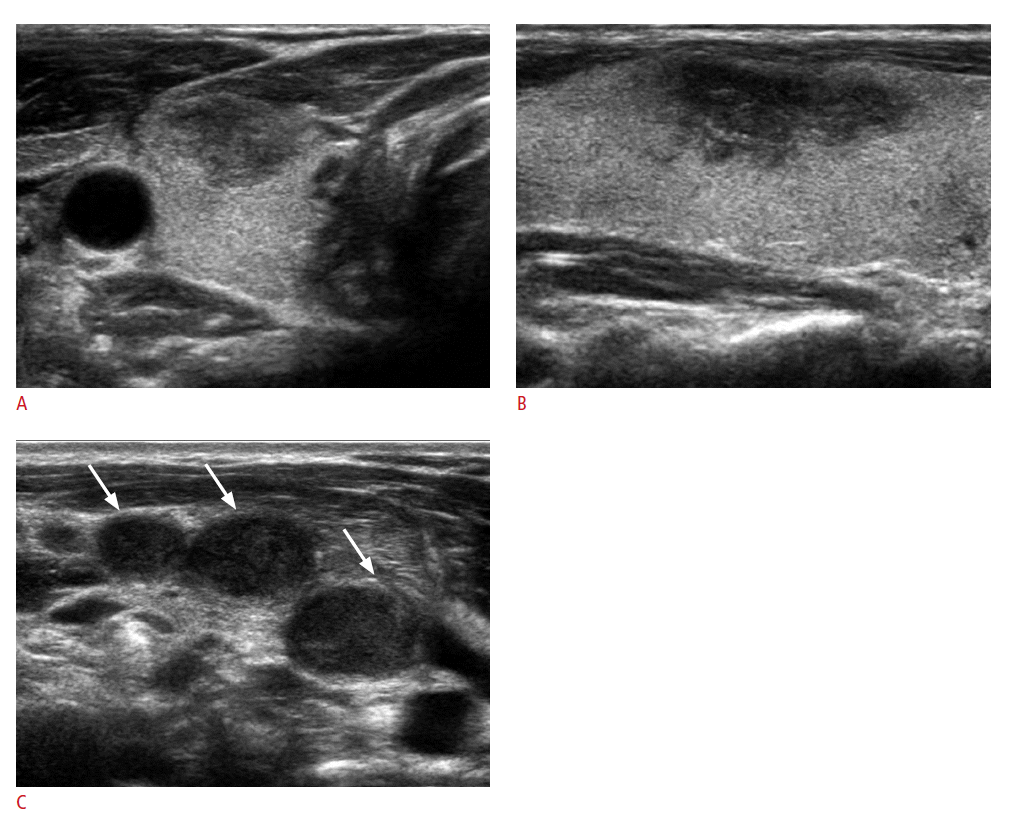
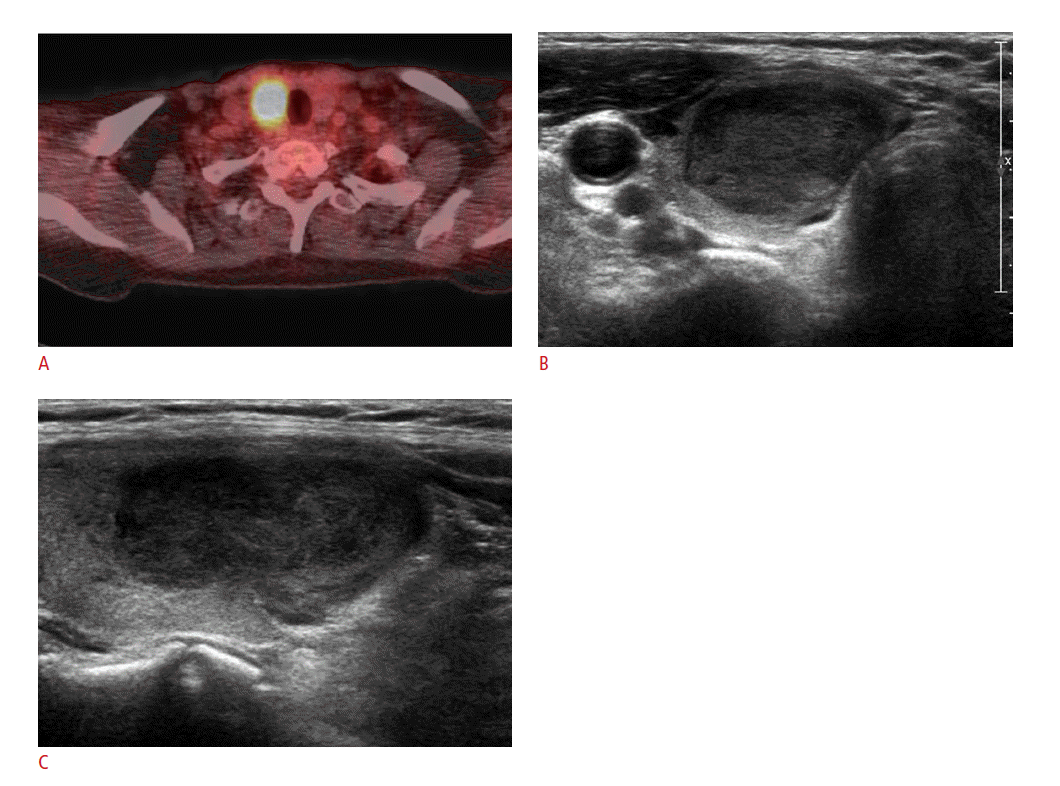
The US features of the 21 mass-forming metastatic lesions are summarized in Table 2. All 21 lesions showed solid tumor composition. Common US features among all 21 thyroid metastases were hypoechogenicity (81.0%); non-circumscribed margins, that is, microlobulated or irregular margins (90.5%); no calcifications (76.2%); and parallel shape (81.0%) (Fig. 2). Of the 14 nodules in which vascularity was available, 12 (85.7%) showed peripheral vascularity on power Doppler US. Regardless of the general US appearance of the 23 thyroid lesions, 21 (91.3%) were classified as suspicious malignancy,, and 2 (8.7%) as probably benign (Fig. 3). Suspicious cervical lymph nodes were present in 18 patients (78.3%) (Figs. 1C, 2C). Three of the 21 suspicious malignant masses had more than 2 suspicious US features, among which hypoechogenicity, irregular margins, and the presence of suspicious cervical lymph nodes were present in all 3 cases. Of the 2 masses that were probably benign, 1 had suspicious cervical lymph nodes, while the other did not show abnormal cervical lymph nodes on US examinations. In the latter case, the thyroid nodule showed intense FDG uptake on PET scans which prompted us to perform US-FNA in spite of rather benign US features.
Of the 23 lesions, 22 had US-FNA performed and 4 had US-CNB done with or without US-FNA. Among the 22 lesions with US-FNA, 21 (95.5%) had diagnostic results specifically suggesting metastasis to the thyroid, and 1 (4.5%) was non-diagnostic. Two had US-CNB performed simultaneously with US-FNA according to the clinician's request for special staining results needed in deciding upon a chemotherapy regimen, and 1 was performed after non-diagnostic cytology for confirmative results.
Discussion
Clinically evident thyroid metastases are considered rather rare, mostly detected only on autopsy [15,21-24]. Technically, all sorts of primary tumors can metastasize to the thyroid, but tumors from the kidney, breast, and lungs were the most frequently reported sites, with clear cell renal cell carcinoma consisting of more than 50% of all thyroid metastases [4,6,25-28]. In our study though, the most common primary tumor site was the lung (37.5%, 9 of 24), followed by the colon and breast. Only 1 patient (4.2%) with renal cell carcinoma had metastasis in the thyroid gland, which is in conflict with many reports. This may have been due to the differences in cancer prevalence among study populations; newly developed malignancies involving the kidney comprise only 2.5% of all cancers, while cancers of the lung, gastrointestinal tract, and breast are leading causes of death in Korea [29].
US features of metastatic carcinoma to the thyroid are known to be nonspecific, appearing as inhomogeneous, hypoechoic, ill-defined, solid lesions with or without metastatic cervical lymphadenopathy [6,11,13,14,26]. The results of our study also revealed similar features; the most common US features in thyroid metastasis were solid composition, hypoechogenicity, non-circumscribed margins, no calcifications, parallel shape, and the presence of suspicious cervical lymph nodes. Most of the lesions included were assessed as suspicious malignancy (91.7%), which may lead to recommending US-FNA for diagnosis, but without acknowledgement of the patient's cancer history, metastatic carcinoma of the thyroid may not be included in the differential diagnosis based on US features alone. Specifically, 17 of the metastatic lesions (73.9%) did not contain microcalcifications, a typical finding representing psammoma bodies in primary thyroid malignancy [30,31]. This, along with other suspicious US features, may lead to suspicion of metastasis in patients with a known primary tumor, as suggested in another study [14]. In addition, 78.3% of the patients included had suspicious cervical lymph nodes present on US examinations. Whereas US features of thyroid metastases prove to be rather nonspecific, the presence of suspicious lymph nodes in a patient who has been diagnosed or treated for cancer may raise suspicion of metastasis in thyroid masses revealed on the same US examination, especially among those showing no suspicious US features. For example, 1 of the 2 cases in our study showing US features that were probably benign had cervical lymph nodes suspicious for metastasis on US (Figs. 1, 2).
Several studies have suggested the preference of metastases to the thyroid in multinodular goiter or pre-existing thyroid nodules [16,26,29,32]. Metastasis to the thyroid is rather rare considering the vast blood volume passing through it, which can be explained by (1) the high oxygen and iodine concentration of the thyroid gland and (2) the rapid arterial blood flow passing through the thyroid gland [7,22,33]. It has been hypothesized that metastatic cells may favor thyroid parenchyma with preexisting thyroid abnormalities such as neoplasms, goiter, or thyroiditis, which contains lower concentrations of iodine or oxygen [16,29], but controversy remains. One report showed the development of metastasis within a preexisting thyroid nodule [16], but the detection of newly developed lesions within the thyroid is generally difficult without serial US examinations performed for comparison. Whether metastases to the thyroid glands actually prefer preexisting thyroid nodules was difficult to evaluate in this study because, in all of the 23 patients, the US examination revealing thyroid metastases was the initial examination performed.
Early detection and treatment of metastases in cancer patients critically affect patient outcomes; therefore, differential diagnosis of thyroid lesions showing these US features is essential. When considering the grave prognosis of the patient if the lesion proves to be metastasis, preoperative diagnosis of the thyroid nodule can help avoid unnecessary surgery [4,6]. As in the differential diagnosis of various thyroid nodules besides metastatic carcinoma, US-FNA can be used as a simple, inexpensive, and effective tool in the cytologic diagnosis of metastatic lesions [3,6,12,19,28,32]. Of the 23 patients included in our study, 22 had undergone US-FNA for diagnosis, among which 21 (95.5%) had been specifically diagnosed with metastasis on cytology. Only 1 patient's cytology turned out to be non-diagnostic, and this patient had thyroid metastasis confirmed by US-CNB. On cytologic evaluation, most cytology specimens from metastatic carcinoma to the thyroid show abundant cellularity with cells typical of the original cancer site [34]. However, even then, without being informed of the possibility of metastasis, diagnosis of metastatic lesions to the thyroid can be rather confusing since the cytology features of metastatic high-grade malignancy are similar to rare primary thyroid malignancies such as anaplastic or clear cell thyroid carcinoma [3,4,6,35]. Additional immunohistochemical stains may also aid the diagnosis of metastasis, showing negative results for thyroglobulin and calcitonin and positive results for cytokeratin subtypes [12,27]. Four patients included in this study had undergone immunohistochemical staining of cytology specimens, all of which showed negative results for thyroglobulin. In spite of the strengths of US-FNA in diagnosis of various thyroid lesions, controversy remains about using US-FNA on nodules suspicious for thyroid metastases, given its high false-negative rates, ranging from 13% to 28.7% [33]. Surgical resection may be considered on selected patients for both diagnosis and complete resection of the metastatic mass, but whether surgery proves to be beneficial in patients with thyroid metastases has not yet been verified.
There are several limitations to our study. First, only one reviewer retrospectively reviewed the US features of the patients included in this study. The observations of additional reviewers may have produced different study results. Second, only 2 patients had been confirmed to have thyroid metastases based on surgical pathology. Although many studies have proven US-FNA to be accurate in the preoperative diagnosis of thyroid metastases, this method has its limitations in differential diagnosis between primary thyroid cancer such as anaplastic or clear cell variants and thyroid metastases [3,4,6,35].
In the present study, most of the thyroid metastases showed nonspecific US features such as hypoechogenicity, non-circumscribed margins, no calcifications, and parallel shape. Therefore, we recommend that US-FNA be performed when a hypoechoic thyroid nodule without malignant US features is sonographically detected in a patient with clinical suspicion of thyroid metastasis.



 Print
Print facebook
facebook twitter
twitter Linkedin
Linkedin google+
google+
 Download Citation
Download Citation PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC




